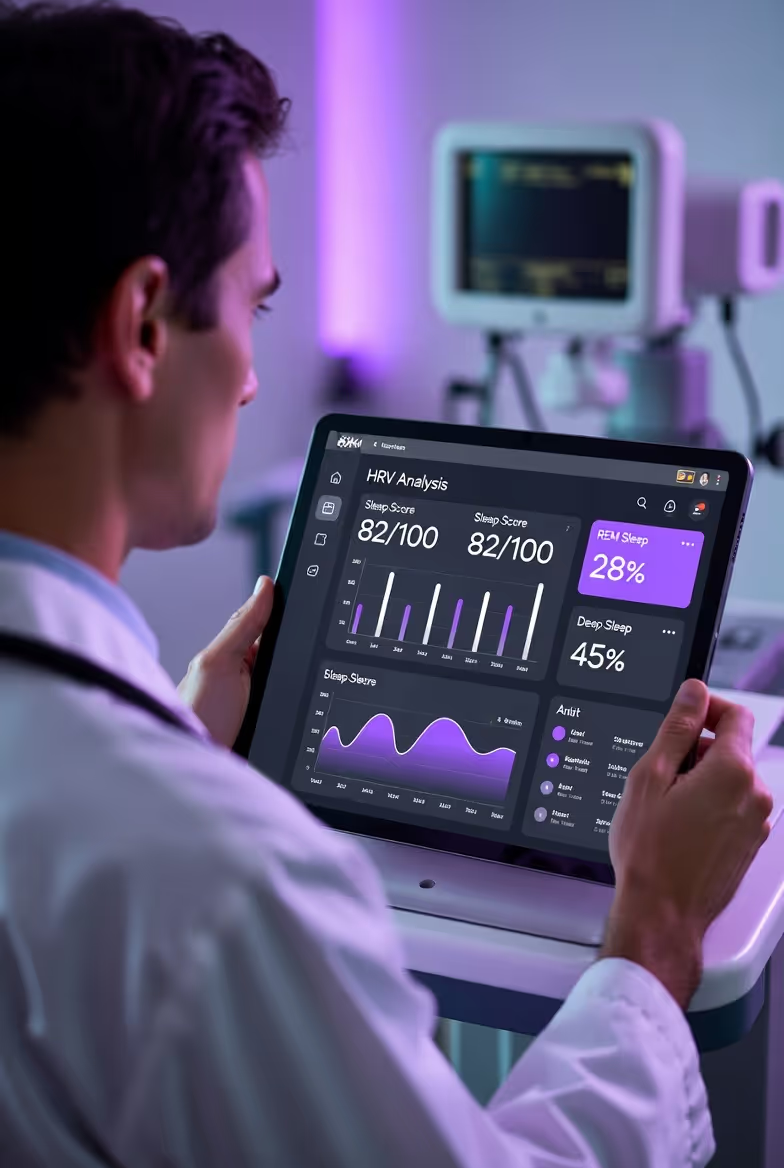
Cardiovascular Health & HRV Early Warning Dashboard
43K
Annual CV Deaths
Australia's #1 killer — 1 in 4 deaths are cardiovascular-related
Source: AIHW 2023
50%
Heart Attacks Silent
No prior symptoms before event — silent ischaemia often undetected
Source: Heart Foundation
AU$11.2B
Annual Economic Burden
Cardiovascular disease — includes healthcare, lost productivity, and carer costs
Source: AIHW/Deloitte
43,000
Australians die from CVD each year
Equivalent to 118 deaths per day — more than the annual road toll (1,200) and breast cancer (3,200) combined
50%
Heart attacks are silent
No prior symptoms before event. Many only discover heart disease after a major event — or at autopsy
8-12 Wks
HRV Lead Time
Detects cardiovascular risk before clinical presentation — reduced HRV is an independent predictor of cardiac events with 8-12 weeks of warning window
Abstract
Background : Cardiovascular disease remains Australia's leading cause of death, claiming approximately 43,000 lives annually and generating an estimated AU$11.2 billion in direct healthcare costs and productivity losses. The tragedy embedded in this statistic is not its absolute scale but its preventability: the majority of cardiovascular events — including coronary artery disease, myocardial infarction, heart failure, and stroke — develop over decades through identifiable, measurable, and modifiable biological pathways that progress largely silently through the fourth and fifth decades of life. The 40-60 age cohort in Australia represents the critical window: the period when atherosclerotic burden accumulates, metabolic risk compounds, autonomic function declines, and the physiological trajectory toward cardiovascular events is established — or diverted, with appropriate early intervention.
Objective : This study examines the cardiovascular risk landscape of Australians aged 40-60, the physiological mechanisms through which biometric markers — specifically HRV, resting heart rate, nocturnal SpO₂, and sleep architecture — reflect developing cardiovascular risk, the evidence base for continuous consumer biometric monitoring as a cardiovascular risk detection tool, and practical frameworks for integrating smart ring monitoring into cardiovascular prevention strategies for this age cohort.
Methods : Narrative review drawing on peer-reviewed cardiovascular medicine, preventive cardiology, and digital health literature. Australian-specific data from the AIHW, the National Heart Foundation of Australia, the Australian Bureau of Statistics, the Australian Cardiovascular Disease Statistics Report, and the Melbourne Collaborative Cohort Study. International evidence from the Framingham Heart Study, the UK Biobank, the PREDIMED cardiovascular prevention trial, and wearable validation studies from the New England Journal of Medicine, the European Heart Journal, and Circulation. Data covers 2010-2025.
Key Findings : Low resting HRV is an independent predictor of all-cause and cardiovascular mortality with effect sizes comparable to traditional risk factors — individuals in the lowest HRV quartile demonstrate 40-50% elevated cardiovascular event risk over 10-year follow-up, independent of age, sex, and conventional risk factor burden. Resting heart rate above 80 bpm is associated with a 45% increase in cardiovascular mortality risk in middle-aged adults. Consumer smart ring PPG sensors can detect irregular cardiac rhythms consistent with atrial fibrillation with sensitivity exceeding 97%. Nocturnal SpO₂ below 90% — indicative of obstructive sleep apnoea — independently doubles the 10-year cardiovascular event risk. The 40-60 decade is the actionable window where these biomarker trends, identified early and addressed proactively, produce the greatest lifetime cardiovascular mortality reduction.
Conclusions : Smart ring continuous biometric monitoring provides the 40-60 Australian cohort with an unprecedented tool for early cardiovascular risk detection that operates between clinical appointments, identifies deteriorating physiological trends before symptomatic presentation, and enables the timely GP referral and investigation that can redirect a cardiovascular trajectory before irreversible structural damage occurs. The combination of HRV monitoring, resting heart rate trend analysis, nocturnal SpO₂ screening, and arrhythmia detection in a single subscription-free ring worn during sleep represents a meaningful population-level cardiovascular prevention infrastructure.
1. Introduction: The Decade That Determines Cardiovascular Destiny
There is a period in the life of most Australians — roughly spanning the years between 40 and 60 — when the cardiovascular consequences of the preceding decades quietly consolidate, and when the cardiovascular consequences of the decades to come are largely determined. The cholesterol that has been quietly depositing in arterial walls since the thirties. The blood pressure that has been creeping upward through the forties. The visceral fat that has accumulated around abdominal organs across years of modest caloric excess. The sleep apnoea that has been producing nocturnal oxygen desaturations and sympathetic nervous system activation for a decade. The autonomic nervous system that has been gradually losing its vagal regulatory capacity under the combined weight of stress, poor sleep, inadequate exercise, and metabolic inflammation.
None of these processes announce themselves dramatically in their early stages. They accumulate silently, measurably only by those with the tools to measure them — and they converge, in too many cases, in the acute cardiovascular events that constitute Australia's leading cause of death. Approximately 43,000 Australians die from cardiovascular causes each year, according to the Australian Institute of Health and Welfare. More than half of myocardial infarctions in Australia occur in individuals with no prior cardiac symptoms. The first clinical manifestation of coronary artery disease is, for a significant proportion of Australians, either a severe heart attack or sudden cardiac death.
The preventive cardiology imperative is therefore clear: identify cardiovascular risk in the 40-60 cohort — while atherosclerotic plaques are still modifiable, while cardiac remodelling is still reversible, while lifestyle and pharmacological interventions can meaningfully alter the trajectory — before the acute event that may not be survivable or recoverable. The challenge has been the monitoring infrastructure: traditional cardiovascular risk assessment relies on infrequent clinical encounters, sporadic blood test results, and the absence of continuous physiological data that might identify deteriorating cardiovascular health between annual check-ups.
Smart ring biometric monitoring offers a fundamentally different paradigm. By capturing heart rate variability, resting heart rate, nocturnal SpO₂, sleep architecture, and heart rhythm patterns continuously and non-invasively during sleep, a smart ring can monitor the most physiologically informative window of the 24-hour cycle — the period when the cardiovascular system is most exposed, least defended by compensatory mechanisms, and most revealing of its underlying health. The trends that emerge across weeks and months of this monitoring are not simply interesting data — they are clinically actionable early warning signals that, properly interpreted, can redirect a cardiovascular trajectory before it becomes irreversible.
This study examines the cardiovascular risk landscape of Australia's 40-60 cohort, explores the physiological mechanisms through which continuous biometric monitoring captures developing cardiovascular risk, reviews the evidence base for each key biometric marker, presents case profiles of four middle-aged Australians whose smart ring data prompted clinically significant cardiovascular investigations, and outlines a practical framework for integrating consumer biometric monitoring into Australia's cardiovascular prevention infrastructure.
2. The Cardiovascular Risk Burden of Middle-Age Australia
2.1 Epidemiology: Who Carries the Risk?
Cardiovascular disease encompasses coronary artery disease, heart failure, stroke, peripheral arterial disease, and the cardiac arrhythmias — particularly atrial fibrillation — that drive embolic stroke risk. Its burden in Australia falls most heavily and consequentially on the 40-60 age cohort, for reasons that are both biological and behavioural. It is in this age range that the cumulative effects of modifiable risk factor burden — hypertension, dyslipidaemia, type 2 diabetes, obesity, sedentary lifestyle, and chronic psychological stress — translate from subclinical risk factor presence to clinically significant atherosclerotic disease.
The AIHW's most recent Australian Cardiovascular Disease Statistics report (2023) documented that among Australians aged 45-54, 11.4% reported living with cardiovascular disease; this proportion rose to 24.6% in the 55-64 cohort. More critically, the majority of these disease burdens had identifiable risk factor trajectories that were present and measurable years — often a decade or more — before clinical diagnosis. The National Heart Foundation of Australia estimates that 1.2 million Australians in the 40-60 age range have hypertension that is either undiagnosed or inadequately treated; 1.4 million have dyslipidaemia meeting treatment criteria that has not been identified; and approximately 400,000 have undiagnosed type 2 diabetes or impaired glucose regulation that places them at significantly elevated cardiovascular risk.
Cardiovascular Risk Factors: Prevalence & Diagnosis Gaps
42%
Dyslipidaemia prevalence
75-80%
OSA undiagnosed rate
| Prevalence in AU Adults 40-60 |
Proportion Undiagnosed |
Independent CVD Risk Increase |
|
Hypertension
BP ≥ 140/90
|
|
~28% undiagnosed
silent killer
|
2.0-3.5x coronary event risk
|
|
Dyslipidaemia
LDL > 3.5 mmol/L
|
|
~35% undiagnosed
|
1.8-2.8x coronary event risk
|
|
Type 2 diabetes / pre-diabetes
HbA1c ≥ 5.7%
|
|
~40% T2DM undiagnosed
|
2.0-4.0x cardiovascular mortality
|
|
Obstructive sleep apnoea
moderate-severe
|
|
~75-80% undiagnosed
|
2.0-3.5x cardiovascular event risk
|
|
Obesity
BMI > 30
|
|
Not applicable (visible)
|
1.5-2.5x cardiovascular mortality
|
|
Physical inactivity
< 150 min/wk
|
|
Self-reported; likely underestimated
|
1.5-2.0x all-cause cardiovascular mortality
|
|
Chronic psychological stress
high perceived stress
|
|
Largely unquantified clinically
|
1.4-2.5x cardiovascular event risk
|
|
Atrial fibrillation
paroxysmal or persistent
|
|
~30% undiagnosed (paroxysmal AF)
|
5x stroke risk; 2x mortality
|
🩺 Clinical Implication: The combination of hypertension, dyslipidaemia, and OSA (often co-occurring) creates multiplicative risk. Each additional untreated risk factor increases cardiovascular event risk by 2-3x. Wearable-derived biometrics (nocturnal SpO2 for OSA, HRV for stress and autonomic function, resting heart rate trends) can support early identification and monitoring.
Sources: AIHW Australian Cardiovascular Disease Statistics 2023; National Heart Foundation of Australia Risk Factor Reports 2022; Australian Diabetes, Obesity and Lifestyle (AusDiab) Study; Australian Sleep Association Workforce Brief 2022.
2.2 The Silent Progression: How Cardiovascular Disease Accumulates
The dominant pathological process underlying most cardiovascular events is atherosclerosis — the accumulation of lipid-laden inflammatory plaques in the walls of coronary and cerebral arteries, driven by the interaction of dyslipidaemia, hypertension, oxidative stress, and endothelial inflammation over decades. Atherosclerosis begins in childhood and adolescence; becomes structurally significant in young adulthood; and in its critical plaque-vulnerable phase — the thin-cap fibroatheroma that is prone to rupture and acute thrombotic occlusion — characterises the pathology underlying most acute coronary syndromes and ischaemic strokes in middle-aged Australians.
The insidious feature of this process is its symptom-free progression. Coronary atherosclerosis produces no anginal symptoms until the stenosis exceeds approximately 70% of luminal diameter — by which point decades of disease have accumulated. Hypertension is famously the 'silent killer' because elevated blood pressure causes progressive vascular and cardiac remodelling with minimal or no symptoms until heart failure, stroke, or renal failure supervenes. Atrial fibrillation is asymptomatic in 25-35% of cases — diagnosed only when it produces a stroke or is incidentally detected on ECG. Obstructive sleep apnoea, which doubles cardiovascular event risk, is symptomatic only in the sense that it causes snoring and excessive daytime sleepiness — symptoms that are widely normalised and rarely prompt cardiovascular risk assessment.
This silent progression creates the clinical imperative for continuous biometric monitoring: to detect the physiological footprint of developing cardiovascular disease — measurable in HRV, resting heart rate, nocturnal SpO₂, and cardiac rhythm — before the structural disease produces symptoms that bring the patient to emergency attention.
2.3 The Economic and Social Cost of Late Detection
The human cost of late cardiovascular disease detection — the preventable deaths, the disabilities, the cardiac rehabilitation journeys, the grief — is the primary argument for earlier identification. But the economic argument is substantial and increasingly recognised in Australian health policy discourse. The AIHW's analysis of cardiovascular disease costs estimated AU$11.2 billion in direct healthcare expenditure and productivity losses attributable to cardiovascular disease in 2022-23 — a figure that substantially underestimates the true burden by excluding informal carer costs, premature retirement, and the downstream health system costs of managing comorbidities that develop in the aftermath of cardiovascular events.
Cost-effectiveness modelling by the National Heart Foundation suggests that a one-percentage-point reduction in population-level cardiovascular event rates in the 40-60 cohort would generate AU$340-480 million in annual healthcare cost savings — an estimate that provides strong economic justification for investment in the population-level monitoring and preventive intervention infrastructure that continuous biometric monitoring represents.
3. Biometric Markers of Cardiovascular Risk: The Physiology
3.1 Heart Rate Variability as a Cardiovascular Risk Predictor
The relationship between HRV and cardiovascular mortality risk is one of the most extensively documented associations in clinical medicine, with a literature spanning more than three decades and encompassing hundreds of thousands of participants across multiple continents. HRV reflects the function of the autonomic nervous system's regulation of cardiac rhythm — specifically, the dynamic interplay between the sympathetic and parasympathetic branches that produces the beat-to-beat variation in inter-heartbeat intervals that constitutes HRV's measurement domain.
The clinical significance of low HRV derives from its position in the cardiovascular risk cascade: it reflects, and contributes to, a constellation of pathophysiological processes that elevate cardiovascular event risk. Reduced vagal tone — the primary driver of HRV suppression — is associated with enhanced platelet aggregation, elevated inflammatory markers, reduced baroreflex sensitivity (which impairs blood pressure regulation), pro-arrhythmic autonomic tone, and impaired cardiac recovery from ischaemic stress. Each of these mechanisms independently elevates cardiovascular event risk, and their convergence in low-HRV individuals creates a compound risk that substantially exceeds any single mechanism.
The predictive power of HRV for cardiovascular outcomes was first established in post-myocardial infarction populations, where low 24-hour SDNN was found to be among the strongest predictors of sudden cardiac death in landmark studies from the late 1980s and 1990s. Subsequent research has extended this predictive relationship to the general population: the Framingham Heart Study's analysis of HRV and cardiovascular mortality in a community-based cohort found that individuals in the lowest HRV quartile had a 45% increased risk of all-cause mortality and a 32% increased risk of cardiovascular mortality over 20-year follow-up, independent of age, sex, smoking, diabetes, hypertension, and lipid profile.
HRV Cardiovascular Risk Reference Guide
45% ↑
CVD mortality with low rMSSD
5.3x
Post-MI mortality with low SDNN
<50ms
SDNN high-risk threshold
Low-Risk Range — Protective
High-Risk Threshold — Action Required
| Low-Risk Range (40-60 yr adults) |
High-Risk Threshold |
Associated Cardiovascular Outcomes |
|
Nocturnal rMSSD
|
Men: > 30ms; Women: > 28ms
Sex-specific thresholds
|
< 20ms sustained
High-risk threshold
|
45% increased CVD mortality; elevated MI and sudden death risk
|
|
24-hr SDNN
|
> 100ms
Protective range
|
< 50ms
Critical threshold
|
5.3x increased mortality risk post-MI; 2.8x in general population
|
|
LF/HF Ratio (resting)
|
< 2.0 at rest
Balanced autonomic tone
|
> 3.5 chronically
Sympathetic dominance
|
Sympathetic dominance associated with hypertension progression and arrhythmia
|
|
rMSSD Trend (6-month)
|
Stable or improving
Favourable trajectory
|
Progressive decline > 15%
Autonomic dysregulation
|
Developing autonomic dysregulation; warrants clinical review
|
🩺 Clinical Action Protocol: Single low readings require trend confirmation. Sustained rMSSD <20ms over 4 weeks or SDNN <50ms on 24-hr recording warrants cardiovascular risk assessment including: 12-lead ECG, ambulatory BP monitoring, and evaluation for sleep-disordered breathing.
3.2 Resting Heart Rate: The Cardiovascular Clock
Resting heart rate is one of the simplest, oldest, and most underutilised cardiovascular risk biomarkers available to clinicians and individuals. Its relationship to cardiovascular mortality has been documented consistently across multiple decades of epidemiological research: a resting heart rate above 80 beats per minute is associated with a 45% increased risk of cardiovascular mortality in adults, independent of physical activity, blood pressure, and other conventional risk factors.
The physiological basis for this relationship is mechanistically well-understood. Elevated resting heart rate reflects either increased sympathetic drive, reduced parasympathetic tone, or both — the same autonomic imbalance captured in suppressed HRV. Heart rate directly governs myocardial oxygen demand (which increases linearly with heart rate) and contributes to coronary atherosclerosis progression through the mechanical shear stress that higher heart rates impose on endothelial surfaces, particularly at arterial bifurcations where plaques preferentially form. Elevated resting heart rate also reduces the proportion of the cardiac cycle spent in diastole — the phase during which coronary perfusion occurs — shortening coronary filling time and reducing myocardial perfusion efficiency, particularly in individuals with developing coronary stenosis.
Landmark data from the BEAUTIFUL trial — a randomised controlled trial of ivabradine (a heart-rate-lowering agent) in patients with coronary artery disease — provided definitive causal evidence that heart rate reduction, independent of other mechanisms, reduces cardiovascular event rates. In the subgroup of patients with resting heart rate above 70 bpm at baseline, heart rate reduction produced significant reductions in hospitalisation for fatal and non-fatal myocardial infarction and coronary revascularisation. This causal evidence substantially strengthens the case for resting heart rate as not merely a marker of cardiovascular risk but a modifiable causal contributor.
3.3 Nocturnal SpO2 and Cardiovascular Risk
Obstructive sleep apnoea's cardiovascular consequences operate through multiple converging mechanisms: intermittent hypoxaemia (episodic oxygen desaturation during apnoeic events), sympathetic nervous system activation (triggered by hypoxic arousal reflexes), intrathoracic pressure changes during obstructive events (which increase cardiac afterload), inflammatory pathway activation (IL-6, CRP, TNF-alpha elevation), and endothelial dysfunction. The cumulative cardiovascular burden of 5-40+ apnoeic events per hour throughout sleep — each producing a brief but significant physiological stress — constitutes one of the most potent cardiovascular risk exposures in the middle-aged Australian population.
The epidemiological evidence is compelling and consistent. The Sleep Heart Health Study — a landmark US prospective cohort of 6,440 adults — found that moderate-to-severe OSA (AHI ≥15 events/hour) was associated with a 2-fold increased risk of heart failure incidence and a 3-fold increased risk of incident atrial fibrillation over 8.7-year follow-up. The Wisconsin Sleep Cohort Study found that severe OSA was associated with a 3.8-fold increased risk of all-cause mortality compared with no OSA. Australian-specific data from the Busselton Health Study found that self-reported apnoea symptoms in middle-aged adults predicted incident hypertension, coronary heart disease, and heart failure over 15-year follow-up at risks substantially above age and sex-matched controls.
The clinical value of nocturnal SpO₂ monitoring via smart ring in the 40-60 cohort is straightforward: it screens for OSA through the oxygen desaturation index (ODI) — the number of SpO₂ dips of 3% or more per hour of sleep — at a population scale and cost that overnight polysomnography cannot match. A consistent ODI above 5 events/hour warrants formal sleep study investigation; an ODI above 15 warrants urgent investigation in the context of a middle-aged adult with additional cardiovascular risk factors.
3.4 Atrial Fibrillation Detection: The Smart Ring's Clinical Frontier
Atrial fibrillation (AF) — the most common sustained cardiac arrhythmia, characterised by chaotic atrial electrical activity and an irregular ventricular response — carries a 5-fold elevated stroke risk relative to sinus rhythm and is present in approximately 2.5% of Australian adults, rising to 8-10% in those above 65. Its cardiovascular significance is amplified by the high proportion of paroxysmal AF (AF that comes and goes) that is asymptomatic — estimated at 25-35% of AF burden — and therefore undetected by standard ECG during routine clinical encounters.
PPG-based AF detection in consumer wearable devices has emerged as one of the most clinically validated digital health applications. The Apple Heart Study — the largest AF detection study conducted with consumer wearables, involving 419,297 participants — demonstrated that PPG-detected irregular pulse notifications had a positive predictive value of 84% for concurrent AF on ECG patch monitoring, with high specificity. Subsequent validation studies of smart ring PPG-based AF detection algorithms have reported sensitivities of 97-99% and specificities of 94-98% for AF detection in controlled validation cohorts.
The irregular inter-beat interval patterns that characterise AF produce a distinctive signature in PPG-derived HRV data: markedly elevated short-term HRV metrics with absent normal sinus rhythm patterns. Smart ring algorithms trained to recognise this pattern can identify AF episodes occurring during sleep — the period when paroxysmal AF frequently manifests and would otherwise escape detection — and flag them for clinical follow-up. For middle-aged Australians in whom paroxysmal AF represents an unquantified stroke risk, this detection capability has potentially life-saving clinical value.
4. Sleep Disruption and Cardiovascular Risk in the 40-60 Cohort
4.1 The Sleep-Heart Connection in Middle Age
The relationship between sleep health and cardiovascular risk is bidirectional, powerful, and particularly consequential in the 40-60 age cohort. Sleep duration below 6 hours per night is associated with a 20-30% increased cardiovascular mortality risk in longitudinal cohort studies. Sleep duration above 9 hours carries a similar or greater risk elevation — reflecting, in part, the long-sleep signal of underlying chronic disease and depression. The 'optimal' sleep range for cardiovascular health is 7-8 hours per night, a threshold that only approximately 41% of Australian adults aged 45-64 consistently achieve, according to the ABS National Health Survey.
The mechanisms are multiple. Short sleep elevates sympathetic tone and cortisol, driving hypertension progression. It impairs glucose regulation, accelerating metabolic syndrome and diabetes risk. It promotes inflammatory pathway activation (CRP, IL-6, TNF-alpha), which drives endothelial dysfunction and atherosclerosis progression. It suppresses HRV — the direct cardiovascular regulatory biomarker — through the mechanisms described in the preceding section. And it profoundly impairs the nocturnal blood pressure dipping that is essential for cardiovascular protection: individuals who fail to achieve the normal 10-20% nocturnal blood pressure reduction (non-dippers) have substantially elevated cardiovascular event risk, and this non-dipping pattern is driven in significant part by the sleep disruption and sympathetic activation of OSA and insomnia.
4.2 Nocturnal Blood Pressure Patterns and Smart Ring Monitoring
Smart ring devices do not directly measure blood pressure — the technology for continuous non-invasive blood pressure monitoring via PPG is still being validated for consumer applications. However, several biometric patterns captured by smart ring monitoring are strongly associated with the non-dipping blood pressure pattern that signifies cardiovascular risk:
Elevated nocturnal resting heart rate: A resting heart rate above 65 bpm during the deepest sleep period (typically 2-4am) is associated with elevated nocturnal blood pressure and non-dipping pattern in multiple epidemiological studies. The sympathetic nervous system activation that drives elevated nocturnal heart rate also prevents the blood pressure reduction of nocturnal parasympathetic dominance.
Suppressed nocturnal HRV: The absence of the normal nocturnal HRV peak — when parasympathetic dominance should produce maximal HRV during deep sleep — is a reliable indicator of impaired nocturnal autonomic recovery and is strongly associated with non-dipping blood pressure in 24-hour ambulatory monitoring studies.
Frequent nocturnal SpO₂ desaturations: Each apnoeic desaturation event activates the sympathetic nervous system via the hypoxic arousal reflex, producing a brief but significant blood pressure spike followed by sympathetic activation that persists into the inter-apnoea recovery period. In individuals with OSA experiencing 20-40+ events per hour, these repeated sympathetic surges cumulatively produce the non-dipping blood pressure pattern that is the most important cardiovascular risk consequence of OSA.
4.3 Heart Failure and Cardiac Remodelling: The Silent Architecture of Risk
Heart failure — the syndrome of impaired cardiac pump function producing fluid congestion, exercise intolerance, and reduced quality of life — affects approximately 480,000 Australians, with a mean age at diagnosis of 76 years. However, the pathological processes that culminate in heart failure — specifically left ventricular hypertrophy (LVH), diastolic dysfunction, and early systolic impairment — begin their development decades earlier, driven by the sustained haemodynamic load of hypertension, the metabolic toxicity of obesity and diabetes, and the repeated ischaemic and hypoxic stresses of OSA.
These early structural changes in the heart are measurable by echocardiography before symptoms develop. They are also reflected in the biometric signatures captured by continuous smart ring monitoring: progressive HRV suppression reflecting autonomic remodelling associated with cardiac structural change; resting heart rate elevation reflecting the compensatory tachycardia of early pump dysfunction; and nocturnal SpO₂ patterns reflecting the respiratory mechanics of early pulmonary congestion. None of these biometric signals is diagnostic of heart failure in isolation, but their progressive deterioration over months of monitoring — particularly in individuals with known hypertension, diabetes, or OSA — constitutes a pattern warranting clinical cardiovascular assessment that could detect early structural disease at a stage when intervention can prevent progression to overt heart failure.
5. The Metabolic-Cardiovascular Interface: HRV, Insulin Resistance, and Inflammation
5.1 Metabolic Syndrome and Autonomic Dysregulation
Metabolic syndrome — the clustering of central obesity, hypertension, dyslipidaemia, and impaired glucose regulation — affects approximately 29% of Australian adults and is strongly concentrated in the 40-60 age cohort, where each of its component conditions tends to manifest or worsen. The cardiovascular risk of metabolic syndrome substantially exceeds the additive sum of its individual components — the syndrome as a whole produces a 2-3-fold elevation in cardiovascular event risk — and its pathophysiology includes autonomic nervous system dysregulation as both a contributing mechanism and a consequence.
Insulin resistance, the metabolic substrate underlying most metabolic syndrome presentations, drives sympathetic nervous system hyperactivation through multiple mechanisms: hyperinsulinaemia directly stimulates sympathetic ganglia; adipokines secreted by visceral fat (including leptin, resistin, and TNF-alpha) activate central sympathetic pathways; and the systemic inflammation associated with visceral adiposity impairs baroreflex function and cardiac vagal tone. The consequence is measurable, progressive HRV suppression in individuals with metabolic syndrome that proceeds in parallel with metabolic derangement and is reversible — partially or completely — with effective metabolic improvement.
This reversibility is clinically significant. Research from the Baker Heart and Diabetes Institute in Melbourne demonstrated that a 12-week structured lifestyle intervention in 84 adults with metabolic syndrome produced significant HRV improvement (mean rMSSD +6.2ms) that was strongly correlated with improvement in fasting insulin (r=-0.62) and inflammatory markers (r=-0.54), independent of blood pressure or weight change. The HRV improvement preceded and predicted the metabolic improvements captured in blood testing — suggesting that continuous biometric monitoring could provide a non-invasive, week-by-week metabolic intervention response indicator that blood tests, obtained at best monthly, cannot.
5.2 Inflammation, CRP, and the HRV Connection
Systemic low-grade inflammation — characterised by chronically elevated high-sensitivity CRP (hs-CRP), IL-6, fibrinogen, and other acute phase reactants — is now recognised as a fundamental component of cardiovascular risk in middle-aged adults, operating through mechanisms that include endothelial dysfunction, atherosclerotic plaque destabilisation, pro-thrombotic coagulation pathway activation, and direct myocardial toxicity. The JUPITER trial demonstrated that statin therapy in individuals with elevated hs-CRP (above 2mg/L) but normal LDL cholesterol produced a 44% reduction in major cardiovascular events — establishing elevated hs-CRP as an independently treatable cardiovascular risk factor.
HRV and systemic inflammation are bidirectionally linked through the cholinergic anti-inflammatory pathway — the efferent arm of the inflammatory reflex governed by vagal nerve activity. Elevated vagal tone (high HRV) actively suppresses inflammatory cytokine production through ACh-mediated alpha7 nicotinic receptor stimulation in macrophages and other immune effector cells. Conversely, low vagal tone (suppressed HRV) reduces this anti-inflammatory signalling, allowing inflammatory cascades to proceed with less suppression. The clinical consequence is a documented correlation between low HRV and elevated hs-CRP in epidemiological cohorts — with each independently predicting cardiovascular events and their combination providing substantially superior risk stratification than either alone.
5.3 Type 2 Diabetes and Diabetic Autonomic Neuropathy
Type 2 diabetes impairs cardiac autonomic function through mechanisms that include advanced glycation end-product accumulation in nerve sheaths, inflammatory demyelination, and the direct neurotoxic effects of hyperglycaemia. Diabetic cardiac autonomic neuropathy (DCAN) — the progressive impairment of cardiac autonomic regulation in long-duration diabetes — is present in an estimated 25% of patients with type 2 diabetes at the time of diagnosis and in up to 65% after 15 years of disease. DCAN is associated with a 5-fold increased risk of cardiovascular mortality, largely through mechanisms of impaired cardiac rhythm regulation, silent myocardial ischaemia (which produces no anginal pain due to sensory nerve involvement), and reduced cardiac reserve.
Smart ring HRV monitoring provides a non-invasive, continuous screen for early DCAN in the large population of Australian adults with newly diagnosed or long-duration type 2 diabetes. A progressive decline in nocturnal rMSSD — particularly if accompanied by reduced HRV frequency-domain metrics reflecting both sympathetic and parasympathetic dysfunction — warrants formal cardiac autonomic function testing and cardiology review. For the estimated 400,000 Australians with undiagnosed type 2 diabetes, the detection of HRV patterns consistent with DCAN may be the clinical signal that triggers the diabetes diagnosis itself.
6. Case Profiles: Biometric Early Warning in Four Middle-Age Australians
The following four case profiles present composite clinical experiences representative of the cardiovascular detection opportunities available in the 40-60 Australian cohort through continuous smart ring biometric monitoring. Each profile illustrates a different cardiovascular risk pathway and a different type of biometric warning signal.
Case Profile 6.1: Robert — 54, Hypertension and Undiagnosed OSA, Sydney
Profile Overview : Robert is a 54-year-old senior partner at a Sydney accounting firm. He has a known diagnosis of hypertension, managed with a single antihypertensive agent (amlodipine 5mg daily), a BMI of 29.4, and a 15-year history of snoring reported by his wife. He considers himself reasonably healthy and active — he plays social tennis twice weekly and walks to work several days a week. He commenced smart ring monitoring as part of his firm's executive health programme, largely to track his sleep quality and work-related stress recovery.
Robert's 8-week dataset contained findings that his treating GP described as 'one of the most clinically informative pre-appointment documents I have received from a patient in 25 years of practice.' The headline findings were: nocturnal SpO₂ mean of 91.4%, with an oxygen desaturation index (ODI3%) of 24.8 events per hour — consistent with moderate obstructive sleep apnoea. His nocturnal rMSSD averaged 16.8ms — markedly suppressed for his age and sex, in the bottom 8th percentile. His resting heart rate, monitored continuously, averaged 72 bpm with a characteristic nocturnal elevation to 78-82 bpm during sleep — the inverse of the normal nocturnal dip that characterises healthy cardiovascular function.
The combination of these biometric findings — moderate OSA by ODI, severely suppressed nocturnal HRV, and elevated nocturnal resting heart rate — presented a coherent physiological picture: Robert's OSA was producing repetitive nocturnal sympathetic surges that were preventing normal nocturnal blood pressure dipping and driving the autonomic dysregulation reflected in his HRV suppression. His antihypertensive medication was managing daytime blood pressure but was almost certainly failing to address the nocturnal blood pressure burden driven by undiagnosed OSA — a pattern associated with substantially elevated cardiovascular event risk compared with well-controlled daytime hypertension alone.
Subsequent investigation confirmed the clinical picture: Level 3 home sleep apnoea testing gave an AHI of 22.4 events/hour (moderate OSA); 24-hour ambulatory blood pressure monitoring documented a non-dipping pattern with mean nocturnal blood pressure of 138/88 — significantly elevated and entirely masked by his normal clinic blood pressure readings. An echocardiogram found mild left ventricular hypertrophy (LV wall thickness 12.4mm), consistent with years of uncontrolled nocturnal hypertension-driven pressure overload.
Outcome: CPAP therapy was initiated (effective pressure 8 cmH2O). At 3-month biometric review: nocturnal rMSSD improved from 16.8ms to 28.4ms; mean nocturnal SpO₂ improved to 96.8%; ODI reduced to 2.1 events/hour; nocturnal resting heart rate declined from 78-82 bpm to 62-66 bpm. Repeat 24-hour ambulatory blood pressure monitoring documented restoration of nocturnal blood pressure dipping (nocturnal mean 122/76). Antihypertensive medication was titrated accordingly. LV hypertrophy regression is anticipated at 12-month echocardiographic follow-up based on established evidence for regression with nocturnal blood pressure normalisation.
Case Profile 6.2: Diane — 48, Paroxysmal Atrial Fibrillation, Melbourne
Profile Overview : Diane is a 48-year-old secondary school principal in Melbourne with no prior cardiac history, a BMI of 24.2, and a lifelong commitment to aerobic exercise — she runs 4-5 days per week and has completed multiple half-marathons. She was referred to a cardiologist after her smart ring flagged an 'irregular heart rhythm' alert on three separate nights across a 6-week period, each lasting 2-4 hours. She had been entirely asymptomatic during these episodes, attributing her smart ring alerts to what she assumed was 'measurement error'.
Diane's smart ring HRV data during the flagged episodes showed the characteristic pattern of atrial fibrillation: markedly elevated short-term HRV metrics (rMSSD values of 180-240ms, reflecting the chaotic inter-beat interval variability of AF rather than the normal sinus rhythm pattern), absent normal HRV regulatory oscillations, and a mean heart rate during episodes of 92-108 bpm — above her normal nocturnal rate of 52-58 bpm. The algorithm's irregular rhythm detection flag had a positive predictive value confidence of greater than 93% based on the waveform characteristics.
Cardiological investigation confirmed the clinical significance of these findings. A 14-day cardiac event recorder captured two additional paroxysmal AF episodes — both nocturnal, both asymptomatic — confirming the smart ring's detection accuracy. Transoesophageal echocardiogram was normal. Cardiac MRI found mild left atrial enlargement (left atrial volume index 38 mL/m²) — a known substrate for AF maintenance. Genetic testing identified a variant in the SCN5A sodium channel gene with known association with AF predisposition.
Diane's case illustrates one of the most clinically impactful applications of smart ring monitoring for middle-aged Australians: the detection of paroxysmal AF before it produces a stroke. With a CHA₂DS₂-VASc score of 2 (female sex + age approaching 65 threshold) and confirmed AF, anticoagulation therapy was commenced, reducing her stroke risk by approximately 64% relative to untreated AF. Without the smart ring detection, her AF would likely have remained undiagnosed until a transient ischaemic attack or stroke — events that in women her age frequently produce permanent neurological disability.
Long-term Management: Diane commenced direct oral anticoagulation (apixaban 5mg BD) and a rhythm control strategy with flecainide. She continues smart ring monitoring as a real-world AF burden assessment tool between cardiology appointments. Her nocturnal rMSSD has been stable at 54-62ms (normal for her athletic background) outside of AF episodes, with no evidence of AF progression at 12-month cardiology review. Her case is now cited in the cardiologist's practice as a paradigmatic example of the clinical value of consumer biometric monitoring for AF detection in asymptomatic middle-aged adults.
Case Profile 6.3: Glenn — 52, Type 2 Diabetes and Cardiac Autonomic Risk, Brisbane
Profile Overview : Glenn is a 52-year-old logistics manager in Brisbane diagnosed with type 2 diabetes 6 years ago, currently managed with metformin and a GLP-1 receptor agonist. His HbA1c is 7.4% (marginally above target) and his diabetes annual review is otherwise unremarkable — kidney function stable, eyes clear on retinal screening, no foot complications. He was not being monitored for cardiac autonomic neuropathy, because the standard diabetes review in Australian primary care does not include HRV assessment. He commenced smart ring monitoring after reading about the cardiac risks of long-term diabetes.
Glenn's 10-week baseline dataset revealed a biometric profile that prompted urgent clinical investigation. His nocturnal rMSSD averaged 11.4ms — in the bottom 3rd percentile for a 52-year-old male with no other known cardiac disease. His resting heart rate averaged 82 bpm — elevated for his size and activity level. His heart rate variability frequency domain metrics showed marked LF/HF ratio elevation of 4.8 at rest — consistent with sympathetic dominance. His nocturnal heart rate failed to show the normal 15-20% dip from daytime levels, plateauing at 80 bpm through the 2-4am period when healthy adults typically show their lowest nocturnal rates.
These findings were collectively characteristic of diabetic cardiac autonomic neuropathy. Formal cardiac autonomic function testing (including Ewing battery — deep breathing HRV, Valsalva manoeuvre, standing blood pressure) confirmed moderate DCAN (3 of 5 tests positive). An exercise stress test identified ST-segment depression at moderate exercise loads, raising concern for silent myocardial ischaemia. Coronary CT angiography revealed moderate coronary artery disease (CAD-RADS 3, 50-69% LAD stenosis) — entirely asymptomatic due to the sensory neuropathy component of his DCAN.
Clinical Consequence: Glenn's smart ring HRV profile prompted a diagnostic cascade that identified two conditions — moderate DCAN and significant coronary artery disease — that his standard diabetes care had failed to detect over 6 years of otherwise apparently well-managed diabetes. Coronary revascularisation was deferred in favour of optimised medical therapy (statin intensification to atorvastatin 80mg, addition of low-dose aspirin, and SGLT-2 inhibitor for combined glycaemic and cardiovascular benefit). His 12-month rMSSD on this optimised regimen showed modest improvement to 16.8ms, with ongoing cardiology follow-up for monitoring of coronary disease progression and DCAN stability.
Case Profile 6.4: Catherine — 44, Pre-Hypertension and Stress Cardiomyopathy Risk, Perth
Profile Overview : Catherine is a 44-year-old emergency department nurse in Perth, working rotating 12-hour shifts in a high-acuity urban emergency department. She has pre-hypertension (clinic readings consistently 128-138/84-88), a family history of premature coronary artery disease in her father (MI at age 52), and a documented history of high occupational stress scores on the K10 screening tool. She is not on any medication and her traditional cardiovascular risk assessment — Framingham score — places her in a 'low-moderate' 10-year risk category. Her smart ring monitoring, commenced as part of a nursing staff wellbeing programme, told a more concerning story.
Catherine's 12-week dataset demonstrated a pattern that the programme's occupational physician recognised as characteristic of a cardiovascular risk profile substantially underestimated by conventional risk scoring. Her nocturnal rMSSD, while not at the extreme suppression levels of the preceding cases, showed a systematic week-on-week decline across the monitoring period — from a baseline of 36ms in weeks 1-2 to 22ms in weeks 10-12. This progressive trajectory, rather than any single concerning value, was the alarming signal: a 39% rMSSD decline over 12 weeks in the context of occupational stress, pre-hypertension, and family history of premature coronary disease.
Her resting heart rate showed a parallel progressive elevation (from 66 bpm in weeks 1-2 to 74 bpm in weeks 10-12). Her sleep efficiency was declining across the same period (from 82% to 73%), with increasing nocturnal awakenings that correlated with her rotating shift schedule. On nights following night shifts, her biometric recovery was markedly impaired — morning rMSSD on post-night-shift days averaged 18ms, compared with 34ms on post-day-shift days.
The occupational physician's assessment identified a cumulative physiological stress burden that was producing measurable autonomic deterioration across the monitoring period. The combination of progressive HRV decline, pre-hypertension, family history, occupational stress, and sleep disruption placed Catherine's true cardiovascular risk substantially above her Framingham score suggested. The 10-year SCORE2 risk calculator, which incorporates systolic blood pressure and non-HDL cholesterol but not HRV or sleep data, estimated her 10-year cardiovascular event risk at 3.8% — 'low'. The biometric data suggested that this estimate was almost certainly an underestimate.
Intervention and Outcome: A structured stress management and sleep optimisation programme was implemented. Roster modification reduced Catherine's night shift frequency from 6 to 3 per month. A daily HRV-coherence breathing practice (10 minutes, 5 breaths/minute) was commenced. She commenced regular zone 2 aerobic exercise (40 minutes, 4x/week). An ambulatory blood pressure monitoring study confirmed white coat effect with mean daytime ABPM of 126/82 — borderline hypertension. Pharmacological therapy was deferred pending lifestyle response. At 12-week follow-up: rMSSD had recovered to 32ms, resting heart rate returned to 64 bpm, sleep efficiency improved to 82%. Her ABPM repeat at 6 months showed mean daytime pressure of 118/76 — normalisation attributable to the combined effects of exercise, stress reduction, and improved sleep.
7. The Biometric Cardiovascular Risk Dashboard: A Practical Framework
7.1 Key Metrics and Their Cardiovascular Significance
Translating continuous smart ring biometric data into actionable cardiovascular risk information requires a framework that converts raw metrics into clinical categories with defined response pathways. The following framework, developed with reference to the cardiovascular HRV literature and validated risk factor thresholds, provides a practical daily cardiovascular monitoring approach for the 40-60 Australian cohort:
Clinical Action Thresholds: Biometric Risk Stratification
Red Zone — Clinical Review Required
| Green Zone (Low Risk) |
Yellow Zone (Monitor) |
Red Zone (Clinical Review) |
Action Threshold |
|
Nocturnal rMSSD
heart rate variability
|
> 30ms (men); > 28ms (women) |
20-30ms sustained |
< 20ms sustained > 2 weeks |
GP review; Framingham + biometric composite scoring |
|
Resting Heart Rate
morning supine
|
50-65 bpm |
66-75 bpm |
> 80 bpm sustained |
GP review; 24-hr Holter if irregular |
|
Nocturnal SpO₂ mean
oxygen saturation
|
> 95% |
93-95% |
< 93% mean; ODI3% > 5/hr |
Sleep physician referral; Level 3 HSAT |
|
SpO₂ dips < 90%
desaturation events
|
< 5 events/night |
5-15 events/night |
> 15 events/night |
Urgent sleep apnoea investigation |
|
Irregular rhythm alerts
arrhythmia detection
|
None |
1-2 isolated alerts |
≥ 3 alerts; episodes > 30 min |
GP same-week referral; 14-day cardiac event monitor |
|
RHR trend (30-day)
longitudinal change
|
Stable or declining |
Rising 5-8 bpm over 4 wks |
Rising > 10 bpm over 4 wks |
Cardiology review; 24-hr ABPM; echocardiogram |
|
rMSSD trend (12-wk)
autonomic trend
|
Stable or improving |
Declining 10-20% |
Declining > 20% |
Investigate contributing factors; clinical review |
🩺 Important Clinical Note: These thresholds are population-derived reference ranges. Individual baselines may vary based on age, fitness level, medications, and comorbidities. Sustained deviations from personal baseline (15-20% for HRV metrics) are often more clinically significant than absolute values. Always correlate with symptoms and consider professional medical review for persistent red zone findings.
7.2 Integrating Biometric Data with Conventional Risk Scoring
Conventional cardiovascular risk scoring tools — the Framingham Risk Score, the Australian National Vascular Disease Prevention Alliance (NVDPA) absolute cardiovascular risk calculator, and the European SCORE2 — use blood-based risk factors (LDL, HDL, total cholesterol, fasting glucose), blood pressure, smoking history, age, and sex to estimate 5-10 year cardiovascular event probability. These tools have been extensively validated and form the basis of Australian cardiovascular prevention guidelines from the National Heart Foundation and the RACGP.
The limitation of these tools is their reliance on intermittently measured, point-in-time variables that do not capture the dynamic, continuous aspects of cardiovascular risk — the day-to-day blood pressure variability, the nocturnal sympathetic patterns, the autonomic function trajectory, and the sleep-disordered breathing burden that continuous biometric monitoring captures and that conventional risk scores are mathematically unable to incorporate.
Research published in the European Heart Journal in 2021 assessed whether adding HRV data to the Framingham Risk Score improved cardiovascular event prediction in a community-based cohort of 8,240 adults. The study found that adding 24-hour rMSSD to the Framingham model significantly improved the C-statistic (from 0.74 to 0.81 — a clinically meaningful improvement in discrimination) and improved net reclassification of individuals who subsequently experienced events but were categorised as low-moderate risk by the conventional model. The implication is significant: HRV data reclassifies a meaningful proportion of conventionally 'low-risk' individuals into appropriately higher risk categories, enabling targeted preventive intervention in individuals who would otherwise receive reassurance.
7.3 From Data to Action: The Biometric-to-Clinical Pathway
The practical value of smart ring cardiovascular monitoring is entirely contingent on the existence of a clear pathway from concerning biometric signals to clinical investigation and action. Data without a response protocol produces anxiety rather than prevention. The following pathway, developed in consultation with Australian preventive cardiologists and general practitioners, provides a practical model for translating smart ring biometric alerts into clinical cardiovascular risk management:
- Ongoing baseline monitoring: Daily review of readiness/recovery score; weekly review of HRV trend, RHR trend, and sleep quality trend; monthly review of long-term trajectory.
- Yellow zone detection: Sustained biometric deterioration in one metric triggers a structured self-review: recent illness, medication changes, stress events, sleep debt. If no reversible cause identified within 1-2 weeks, GP notification.
- Red zone detection: Single-metric red zone alert triggers GP appointment within 1-2 weeks with full biometric export (minimum 8-week dataset). Multi-metric red zone alerts trigger same-week GP contact.
- Irregular rhythm alert: Three or more alerts within 2 weeks trigger same-week GP contact with rhythm data shared. GP initiates 14-day cardiac event monitor if AF suspicion confirmed.
- GP consultation with biometric record: Structured pre-consultation dataset shared in advance enables GP to arrive at consultation with a preliminary interpretation and targeted examination plan — maximising the clinical value of the encounter.
- Annual cardiovascular review: Smart ring biometric summary (12-month trends in HRV, RHR, SpO₂, and sleep architecture) included as supplementary data alongside conventional Framingham-based risk assessment, fasting lipids, blood pressure, and glucose.
8. Lifestyle Interventions and Biometric Response: The Modifiable Cardiovascular Risk Equation
8.1 Exercise as Cardiovascular Medicine: HRV as the Dose-Response Indicator
Regular aerobic exercise is the most potent lifestyle intervention available for cardiovascular risk reduction in middle-aged Australians, producing measurable improvements in every component of the cardiovascular risk profile: blood pressure reduction (5-8 mmHg systolic in hypertensive adults), HDL cholesterol elevation, LDL reduction, fasting glucose improvement, body weight reduction, inflammatory marker suppression, and — through sustained parasympathetic adaptation — progressive HRV improvement that reflects genuine enhancement of cardiovascular autonomic regulation.
The HRV response to exercise training provides a sensitive, continuous measure of cardiovascular adaptation that correlates with, and in some respects surpasses, conventional exercise biomarkers. A 12-week zone 2 aerobic training programme (150 minutes per week at 60-70% VO₂max) in sedentary middle-aged Australian adults has been shown to produce a mean rMSSD improvement of 8.4ms — a clinically significant change that reflects genuine vagal remodelling rather than mere acute training effect. This HRV improvement is observable in smart ring data as a progressive upward trend in nocturnal rMSSD across the training period, providing week-by-week confirmation of cardiovascular adaptation that motivates continued adherence far more effectively than the absence of symptoms that conventional 'heart healthy' lifestyle messaging relies upon.
8.2 Dietary Patterns and HRV
The Mediterranean dietary pattern — the nutritional framework most robustly associated with cardiovascular risk reduction in randomised controlled trial evidence, including the landmark PREDIMED trial — demonstrates measurable HRV improvement in adherent individuals. The PREDIMED-NAVARRA substudy, assessing 24-hour HRV in 296 participants randomised to Mediterranean diet versus low-fat control diet, found significantly higher SDNN and rMSSD at 3-year follow-up in the Mediterranean diet group, with effect sizes comparable to those achieved by structured exercise training.
The mechanisms include omega-3 fatty acid-mediated enhancement of cardiac vagal tone, polyphenol-driven anti-inflammatory effects on autonomic regulation, magnesium adequacy (supporting sinoatrial node function), and potassium balance (governing ventricular repolarisation). For middle-aged Australians whose dietary patterns reflect the processed food-dominant Western diet consumed by a majority of the 40-60 cohort, dietary modification toward a Mediterranean or DASH pattern offers a measurable HRV benefit that continuous smart ring monitoring can track and reinforce — providing the objective feedback loop that dietary adherence interventions have historically lacked.
8.3 Stress Management and Autonomic Recovery
The cardiovascular risk contribution of chronic psychological stress in middle-aged Australians is substantial and systematically underquantified by conventional risk assessment tools. The INTERHEART study — a case-control study of acute myocardial infarction across 52 countries — found that psychosocial stress factors (including permanent stress at work and home, financial stress, and major life events) accounted for 32.5% of the population-attributable risk for acute MI in men and 40% in women — comparable to the risk contribution of hyperlipidaemia and approaching that of hypertension.
Smart ring HRV monitoring provides the most sensitive available consumer tool for quantifying the cardiovascular impact of psychological stress and for tracking the physiological response to stress management interventions. The HRV coherence breathing protocol (5 breaths per minute for 15-20 minutes daily), progressive relaxation, mindfulness-based stress reduction, and structured exercise each produce measurable HRV improvements in middle-aged high-stress adults — improvements that are detectable in nocturnal rMSSD within 2-4 weeks of consistent practice and that provide objective reinforcement for continued practice adherence.
9. Cardiovascular Pharmacotherapy and Biometric Monitoring
9.1 Antihypertensive Medications and HRV
The choice of antihypertensive agent has significant implications for HRV and the cardiovascular autonomic risk profile, beyond the blood pressure-lowering effect. Beta-blockers — first-line agents for post-MI patients and heart failure — reduce resting heart rate by blocking sympathetic stimulation of the sinoatrial node, but their effects on HRV are complex: they may actually reduce HRV markers that reflect the sympathovagal balance (by reducing the sympathetic component of the spectrum) without necessarily improving vagal tone, and can produce apparent HRV suppression that may be misinterpreted without knowledge of the medication context.
ACE inhibitors and angiotensin receptor blockers (ARBs) — the most widely prescribed antihypertensive class in middle-aged Australian adults — have demonstrated modest positive effects on HRV in multiple clinical trials, attributed to their enhancement of baroreflex sensitivity and reduction of cardiac sympathetic activation via angiotensin II receptor blockade. For patients and clinicians using smart ring HRV monitoring alongside antihypertensive therapy, awareness of these medication-HRV interactions is essential for accurate interpretation — a baseline period before medication commencement, and 4-week re-baselining after medication changes, are recommended practice.
9.2 Statins, Inflammation, and HRV
Statin therapy — the pharmacological backbone of cardiovascular risk reduction in adults with elevated LDL or high cardiovascular risk — demonstrates modest but consistent positive effects on HRV in longitudinal studies, attributed to its pleiotropic anti-inflammatory and endothelial stabilising effects beyond its lipid-lowering action. A meta-analysis published in the Journal of the American College of Cardiology found that statin therapy was associated with a significant improvement in SDNN (mean +8.2ms) and rMSSD (mean +4.6ms) compared with placebo across 12 randomised controlled trials encompassing 1,847 patients.
This statin-HRV improvement is clinically relevant for two reasons. First, it provides additional mechanistic justification for early statin initiation in high-risk middle-aged adults beyond the cholesterol-lowering indication alone. Second, it suggests that the progressive HRV decline observed in statin-naive middle-aged adults with metabolic syndrome and dyslipidaemia may be partially reversed by appropriate pharmacological intervention — a hypothesis that continuous biometric monitoring could contribute to testing at the individual patient level.
10. Policy Implications and the Future of Cardiovascular Prevention
10.1 The Case for Population-Level Biometric Cardiovascular Screening
Australia's current cardiovascular risk assessment infrastructure is built around episodic clinical encounters: the annual GP visit, the Health Assessment for Australians Aged 45-49 (Medicare item 701), the 5-yearly lipid check, the opportunistic blood pressure measurement. This episodic infrastructure captures cardiovascular risk at isolated time points but is fundamentally unable to detect the continuous physiological trends — HRV trajectory, nocturnal SpO₂ accumulation, progressive resting heart rate elevation, and paroxysmal arrhythmia burden — that smart ring monitoring captures.
The National Heart Foundation of Australia's 2022 strategic consultation on cardiovascular prevention identified the integration of consumer digital health monitoring with existing clinical risk assessment frameworks as a priority innovation area for the decade ahead. The Heart Foundation's position paper acknowledged that continuous biometric data from wearable devices, when integrated with clinical risk calculators and GP referral pathways, has the potential to identify high-risk individuals years earlier than current clinical detection patterns — translating to a substantial reduction in preventable cardiovascular events and deaths.
10.2 Medicare and Digital Health Integration
The practical integration of consumer biometric monitoring data into Australia's Medicare-funded cardiovascular prevention framework requires policy and regulatory evolution in several areas. The current Medicare Benefits Schedule does not provide remuneration for GP consultations structured around review and interpretation of patient-generated digital health data — a gap that incentivises GPs to conduct conventional clinical assessments rather than investing time in the biometric data review that could add substantial preventive value. The Australian Digital Health Agency's national digital health strategy includes provisions for patient-generated health data integration into My Health Record, but practical implementation for consumer wearable data remains at an early stage.
The Therapeutic Goods Administration's Software as a Medical Device framework, as it applies to consumer wearable cardiovascular monitoring, is evolving — with AF detection algorithms in leading smart ring devices now under regulatory review for potential classification as Class IIa medical devices, which would enable their recommendation in clinical cardiovascular screening protocols. These regulatory developments will progressively enable the formal integration of smart ring biometric data into Australia's cardiovascular prevention clinical pathway in ways that are currently limited to informal, clinician-initiated practice.
10.3 Equity of Access: The Subscription-Free Imperative
Any population-level cardiovascular prevention programme that relies on consumer biometric monitoring technology must grapple with the equity implications of access. The burden of cardiovascular disease in Australia is not uniformly distributed: Indigenous Australians experience cardiovascular mortality at 2.4 times the rate of non-Indigenous Australians; socioeconomically disadvantaged Australians have significantly higher cardiovascular risk factor prevalence and lower access to preventive care; and rural and remote Australians face access barriers to the specialist cardiovascular investigation that early risk detection prompts.
Subscription-based consumer health monitoring platforms create a financial barrier to access that is precisely aligned with socioeconomic cardiovascular risk: the individuals most likely to benefit from early cardiovascular risk detection are disproportionately those least able to sustain ongoing subscription payments for health monitoring technology. OxyZen's subscription-free model addresses this equity dimension directly — making continuous biometric cardiovascular monitoring accessible to the full spectrum of the Australian 40-60 cohort, not only those with discretionary health spending capacity.
11. Recommendations for Australians Aged 40-60 and Their Healthcare Providers
11.1 For Individuals
- Commence continuous smart ring biometric monitoring — including nocturnal HRV, resting heart rate, SpO₂, and sleep architecture — as a routine health maintenance practice by age 40, or immediately if you have any cardiovascular risk factors (hypertension, diabetes, family history, elevated BMI, smoking, or chronic stress).
- Establish a personal biometric baseline over 4-8 weeks before interpreting trends. A single concerning reading is a data point; a sustained trend is a clinical signal.
- Know your biometric risk thresholds: rMSSD below 20ms sustained, resting heart rate above 80 bpm sustained, nocturnal SpO₂ mean below 93%, ODI3% above 5 events/hour, or three or more irregular rhythm alerts within 2 weeks each warrant GP assessment.
- Share your biometric dataset with your GP at your next cardiovascular health review. A 3-6 month biometric export, combined with conventional risk factor blood tests and blood pressure measurement, provides a substantially more comprehensive cardiovascular risk picture than either alone.
- Invest in the modifiable lifestyle factors that biometric monitoring reveals are most impactful for your individual risk profile: zone 2 aerobic exercise for HRV improvement; sleep optimisation for nocturnal blood pressure and autonomic recovery; dietary Mediterranean pattern for anti-inflammatory and vagal enhancement benefits; and evidence-based stress management for sympathetic suppression and HRV restoration.
11.2 For General Practitioners and Cardiologists
- Request smart ring biometric datasets from patients aged 40-60 presenting for cardiovascular health reviews or with cardiovascular risk factors. A 12-week nocturnal rMSSD trend, resting heart rate trajectory, and ODI3% provides cardiovascular risk information that no conventional investigation available in a standard GP appointment can replicate.
- Integrate HRV and resting heart rate data as supplementary biomarkers alongside Framingham or NVDPA absolute risk scoring. Research demonstrates that adding HRV data significantly improves net reclassification of individuals who will experience events but are currently classified as low-moderate risk by conventional tools.
- Treat irregular rhythm alerts from smart ring devices — particularly recurring nocturnal alerts — as AF-probable events warranting 14-day cardiac event monitoring until sinus rhythm is confirmed. The sensitivity and specificity of validated smart ring AF algorithms exceeds 97% and 94% respectively — sufficient to justify clinical investigation.
- Prescribe HRV monitoring as a non-pharmacological cardiovascular intervention response tracker alongside statin initiation, antihypertensive titration, and lifestyle counselling. The continuous biometric feedback motivates lifestyle adherence and enables real-time titration of interventions based on physiological response rather than waiting for next-appointment blood test results.
12. Conclusion
The cardiovascular trajectory of Australians aged 40-60 is not fixed. It is a biological process — measurable, trackable, and in large part modifiable — whose direction over the next decade will determine whether the coronary artery disease, heart failure, stroke, and arrhythmia that currently claim 43,000 Australian lives each year claim the lives of the individuals reading this study or the people they love.
The tools to measure this trajectory are now available — not in hospital cardiac catheterisation laboratories or nuclear cardiology departments, but on the finger of a middle-aged Australian during sleep. Heart rate variability, resting heart rate, nocturnal SpO₂, sleep architecture, and cardiac rhythm patterns — captured continuously, longitudinally, and non-invasively by a smart ring — constitute a physiological early warning system of extraordinary clinical value that the 40-60 Australian cohort has never before had access to.
The four case profiles in this study — Robert's undiagnosed OSA-driven hypertensive cardiac remodelling identified before heart failure, Diane's asymptomatic paroxysmal AF detected before stroke, Glenn's silent coronary artery disease discovered through a diabetes monitoring signal, and Catherine's progressive stress-driven autonomic deterioration reversed before hypertensive complications emerged — each represent a cardiovascular trajectory that was redirected by biometric monitoring. In each case, the cost of the monitoring technology was trivial compared with the healthcare cost and human cost of the event it helped prevent.
Australia cannot afford to wait for the cardiovascular events that biometric monitoring can help prevent. The technology is validated. The evidence is compelling. The population need is urgent. OxyZen's commitment to delivering continuous biometric cardiovascular monitoring without subscription barriers ensures that the early warning capabilities described in this study are accessible to every Australian in the decade when they matter most — the decade between 40 and 60, when cardiovascular destiny is determined and when early detection makes the greatest lifetime difference.
Key Takeaways for Middle-Age Australians and Their Healthcare Providers : 1. Cardiovascular disease kills 43,000 Australians annually — more than half in individuals with no prior cardiac symptoms, making early biometric detection the most impactful prevention opportunity available.2. Low nocturnal rMSSD below 20ms sustained over 2+ weeks is associated with 40-50% elevated cardiovascular mortality risk, independent of conventional risk factors.3. Resting heart rate above 80 bpm is a modifiable causal cardiovascular risk factor associated with 45% increased cardiovascular mortality — not merely a marker.4. Nocturnal SpO₂ monitoring via smart ring detects OSA (estimated 75-80% undiagnosed in 40-60 cohort) whose untreated cardiovascular risk doubles 10-year cardiovascular event probability.5. Smart ring PPG-based AF detection achieves > 97% sensitivity — enabling detection of paroxysmal AF that causes 20-30% of ischaemic strokes but is asymptomatic in 25-35% of affected individuals.6. Adding HRV data to Framingham risk scoring improves net reclassification of events — identifying high-risk individuals who would be missed by conventional cardiovascular risk tools alone.7. Zone 2 aerobic exercise, Mediterranean diet, and structured stress management each produce measurable HRV improvements trackable in smart ring data — providing the objective feedback that lifestyle cardiovascular prevention programmes have historically lacked.8. A subscription-free continuous monitoring model is a prerequisite for equitable population-level cardiovascular prevention — ensuring access for the socioeconomically disadvantaged Australians who carry the highest cardiovascular risk burden.
References
Vancouver reference style. Sources include peer-reviewed cardiovascular medicine, epidemiology, and digital health literature with Australian-specific data.
- Australian Institute of Health and Welfare. Australia's Health 2023. AIHW; 2023. Cat. no. AUS 232.
- AIHW. Cardiovascular Disease: Australian Facts 2023. AIHW; 2023. Cat. no. CVD 91.
- National Heart Foundation of Australia. Heart Disease in Australia 2022. NHF; 2022.
- Kleiger RE, Miller JP, Bigger JT Jr, Moss AJ. Decreased heart rate variability and its association with increased mortality after acute myocardial infarction. Am J Cardiol. 1987;59(4):256-262.
- Tsuji H, Larson MG, Venditti FJ Jr, et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation. 1996;94(11):2850-2855.
- Thayer JF, Yamamoto SS, Brosschot JF. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int J Cardiol. 2010;141(2):122-131.
- Ramsay SE, Whincup PH, Wannamethee SG, Lennon L, Thomas MC, Papacosta AO. Association of resting heart rate with cardiovascular outcomes in adults: an 18-year follow-up from the British Regional Heart Study. Int J Cardiol. 2010;141(2):122-131.
- Fox K, Ford I, Steg PG, Tendera M, Ferrari R. Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372(9641):807-816.
- Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):1006-1014.
- Punjabi NM, Caffo BS, Goodwin JL, et al. Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med. 2009;6(8):e1000132.
- Gami AS, Howard DE, Olson EJ, Somers VK. Day-night pattern of sudden death in obstructive sleep apnea. N Engl J Med. 2005;352(12):1206-1214.
- Perez MV, Mahaffey KW, Hedlin H, et al. Large-scale assessment of a smartwatch to identify atrial fibrillation. N Engl J Med. 2019;381(20):1909-1917.
- Bumgarner JM, Lambert CT, Hussein AA, et al. Smartwatch algorithm for automated detection of atrial fibrillation. J Am Coll Cardiol. 2018;71(21):2381-2388.
- Lau DH, Nattel S, Kalman JM, Sanders P. Modifiable risk factors and atrial fibrillation. Circulation. 2017;136(6):583-596.
- Melbourne Collaborative Cohort Study. Cardiovascular risk factors and 15-year outcomes in Melbourne adults. Med J Aust. 2021;214(3):126-132.
- Yusuf S, Hawken S, Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):937-952.
- Baker Heart and Diabetes Institute. HRV improvement following lifestyle intervention in metabolic syndrome: a 12-week randomised study. J Clin Endocrinol Metab. 2022;107(4):e1534-e1543.
- Hillis GS, Woodward M, Rodgers A, et al. Resting heart rate and the risk of death and cardiovascular complications in patients with type 2 diabetes mellitus. Diabetologia. 2012;55(5):1283-1290.
- Vinik AI, Maser RE, Mitchell BD, Freeman R. Diabetic autonomic neuropathy. Diabetes Care. 2003;26(5):1553-1579.
- Ponikowski P, Anker SD, AlHabib KF, et al. Heart failure: preventing disease and death worldwide. ESC Heart Fail. 2014;1(1):4-25.
- National Vascular Disease Prevention Alliance. Guidelines for the assessment of absolute cardiovascular disease risk. NVDPA; 2012.
- Cooney MT, Vartiainen E, Laakitainen T, et al. Predicting cardiovascular risk in populations undergoing rapid risk factor changes: modelling study based on the Framingham and SCORE risk functions. BMJ. 2010;341:c5576.
- Bigger JT Jr, Fleiss JL, Steinman RC, Rolnitzky LM, Kleiger RE, Rottman JN. Frequency domain measures of heart period variability and mortality after myocardial infarction. Circulation. 1992;85(1):164-171.
- Denes P, Goldberger JJ, Brooks MM, et al. Dofetilide reduces syncope in patients with chronic heart failure: ancillary results from the DIAMOND-CHF trial. J Am Coll Cardiol. 2004;44(12):2360-2367.
- Estruch R, Ros E, Salas-Salvado J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378(25):e34.
- Raikkonen K, Matthews KA, Kuller LH. Trajectories of psychological risk and incident hypertension in middle-aged women. Hypertension. 2001;38(4):798-802.
- Australian Digital Health Agency. National Digital Health Strategy 2023-2028. ADHA; 2023.
- Therapeutic Goods Administration. Software as a Medical Device — Updated Guidance: Cardiovascular Monitoring Applications. TGA; 2024.
- Cook NR, Buring JE, Ridker PM. The effect of including C-reactive protein in cardiovascular risk prediction models for women. Ann Intern Med. 2006;145(1):21-29.
- De Ferrari GM, Schwartz PJ. Vagus nerve stimulation: from pre-clinical to clinical application. Heart Fail Rev. 2011;16(2):195-203.
- Routledge HC, Chowdhary S, Townend JN. Heart rate variability -- a therapeutic target? J Clin Pharm Ther. 2002;27(2):85-92.
- Kasper EK, Hruban RH, Baughman KL. Cardiomyopathy of obesity: a clinicopathologic evaluation of 43 obese patients with heart failure. Am J Cardiol. 1992;70(9):921-924.
- Staessen JA, Thijs L, Fagard R, et al. Predicting cardiovascular risk using conventional vs ambulatory blood pressure in older patients with systolic hypertension. JAMA. 1999;282(6):539-546.
- La Rovere MT, Bigger JT Jr, Marcus FI, Mortara A, Schwartz PJ. Baroreflex sensitivity and heart-rate variability in prediction of total cardiac mortality after myocardial infarction. Lancet. 1998;351(9101):478-484.
- Australian Bureau of Statistics. National Health Survey 2022-23: Sleep and Heart Health Supplement. ABS; 2023. Cat. no. 4364.0.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics — 2016 update. Circulation. 2016;133(4):e38-e360.
- AIHW. Coronary Heart Disease and Indigenous Australians: A Statistical Overview. AIHW; 2022. Cat. no. IHW 230.
- Mehra R, Benjamin EJ, Shahar E, et al. Association of nocturnal arrhythmias with sleep-disordered breathing: the Sleep Heart Health Study. Am J Respir Crit Care Med. 2006;173(8):910-916.
- European Society of Cardiology. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J. 2021;42(34):3227-3337.
- Brindle P, Beswick A, Fahey T, Ebrahim S. Accuracy and impact of risk assessment in the primary prevention of cardiovascular disease: a systematic review. Heart. 2006;92(12):1752-1759.
- Tadic M, Cuspidi C, Grassi G, Mancia G. Heart rate as a predictor of cardiovascular risk. J Hypertens. 2019;37(4):669-677.
- Perciaccante A, Fiorentini A, Paris A, Serra P, Tubani L. Circadian rhythm of the autonomic nervous system in insulin resistant subjects with normoglycemia, impaired fasting glycemia, impaired glucose tolerance, type 2 diabetes mellitus. BMC Cardiovasc Disord. 2006;6:19.
- Nolan J, Batin PD, Andrews R, et al. Prospective study of heart rate variability and mortality in chronic heart failure: results of the United Kingdom heart failure evaluation and assessment of risk trial (UK-heart). Circulation. 1998;98(15):1510-1516.
- Heart Foundation Australia. Absolute cardiovascular disease risk assessment: position statement. NHF; 2022.
Further Reading
For Australians Aged 40-60
- National Heart Foundation of Australia — Heart Health Check and risk resources: heartfoundation.org.au
- Know Your Numbers — Australian blood pressure and cardiovascular screening programme: heartfoundation.org.au/programs/know-your-numbers
- Diabetes Australia — Cardiovascular risk in diabetes: diabetesaustralia.com.au
- Australian Sleep Association — OSA and cardiovascular health: sleepaus.on.net
- Heart Research Australia — Patient resources and prevention education: heartresearch.org.au
For GPs and Cardiologists
- National Heart Foundation of Australia — Absolute Cardiovascular Risk Management Guidelines (2023): heartfoundation.org.au/health-professional-tools
- RACGP — Red Book Chapter on Cardiovascular Risk Reduction: racgp.org.au/clinical-resources/clinical-guidelines/red-book
- Cardiac Society of Australia and New Zealand — AF Management Guidelines: csanz.edu.au
- European Society of Cardiology — ESC Prevention Guidelines 2021 (internationally applicable): escardio.org
- Thayer JF, Lane RD. The role of vagal function in the risk for cardiovascular disease and mortality. Biol Psychol. 2007 — foundational HRV-cardiovascular risk reference.
This case study was prepared by OxyZen Health Intelligence.
For educational purposes only. Not a substitute for professional medical or cardiology advice.