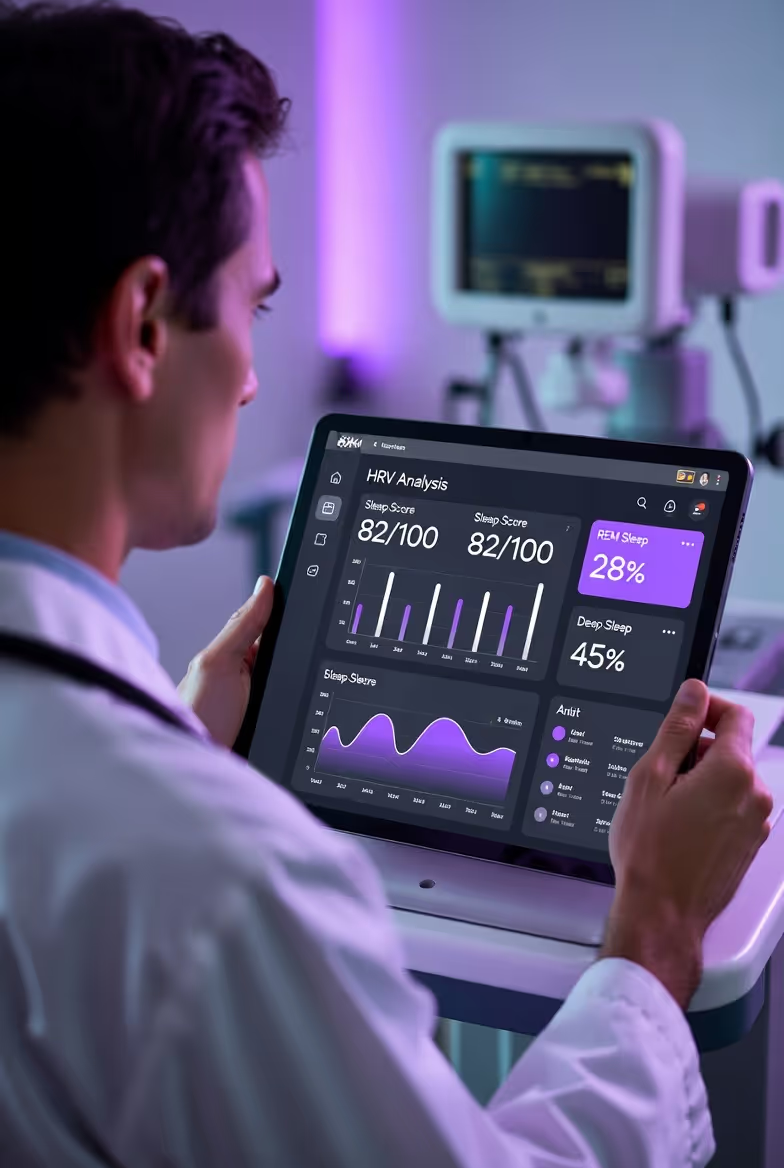
Women's Health & Cycle Tracking Dashboard
6.1M
Menstruating Australians
Aged 15-54 (ABS 2021) — representing 24% of the total population
Source: Australian Bureau of Statistics
0.3°C
BBT Shift at Ovulation
Detectable via smart ring sensor — the most reliable physiological marker of ovulation
Source: Fertility tracking studies
71%
Cycle Irregularity Linked
To identifiable hormonal disorder — PCOS, endometriosis, thyroid dysfunction, or hyperprolactinaemia
Source: RANZCOG Guidelines
1 in 9
Women with Endometriosis
Affecting approximately 830,000 Australian women — equivalent to the population of Adelaide
⏱️ Average 7-year diagnosis delay from symptom onset
Abstract
Background : The menstrual cycle is one of the most information-rich biological signals in the human body. Across its 21-35 day length, the interplay of oestrogen, progesterone, luteinising hormone, and follicle-stimulating hormone orchestrates measurable changes in basal body temperature, heart rate variability, resting heart rate, respiratory rate, sleep architecture, skin temperature, and autonomic nervous system tone — changes that are precisely time-stamped by ovulation and the luteal-follicular transition. For the approximately 6.1 million menstruating Australians aged 15-54, and the millions more navigating perimenopause and post-reproductive hormonal transition, these physiological signals contain clinically valuable information about reproductive health, hormonal status, and systemic wellbeing that has historically been invisible — accessible only through expensive laboratory hormone testing or the subjective self-observation methods of traditional fertility awareness.
Objective : This study examines the hormonal physiology of the menstrual cycle and how its biomarkers are captured by smart ring wearable technology; reviews the evidence base for temperature-based and HRV-based cycle tracking; explores the clinical value of continuous biometric monitoring for women with PCOS, endometriosis, perimenopause, and fertility challenges; and presents case profiles of four Australian women whose hormonal health journeys were meaningfully informed by smart ring biometric data.
Methods : Narrative review of reproductive endocrinology, menstrual cycle physiology, wearable technology validation studies, and women's health epidemiological data. Sources include Jean Hailes for Women's Health Australia, the Australasian Menopause Society, the PCOS Australia Alliance, Endometriosis Australia, the Medical Journal of Australia, the Journal of Women's Health, Human Reproduction, Fertility and Sterility, and research from Monash University's Department of Obstetrics and Gynaecology, the University of Adelaide's Robinson Research Institute, and the University of New South Wales. Data covers 2010-2025.
Key Findings : Smart ring distal skin temperature sensors detect the post-ovulatory basal body temperature rise of 0.2-0.5 degrees Celsius with accuracy comparable to traditional BBT thermometry when measurements are standardised to stable overnight conditions. Nocturnal HRV demonstrates a robust, reproducible luteal-phase suppression of 8-14% compared with follicular-phase baseline across multiple validated studies. Women with PCOS demonstrate significantly blunted HRV cyclicity and higher resting heart rates than eumenorrhoeic controls, providing a non-invasive biomarker of the sympathetic hyperactivation characteristic of the syndrome. Women in the perimenopausal transition show measurable HRV decline and increased temperature instability that precedes and accompanies vasomotor symptoms, offering an objective monitoring window for hormonal treatment decisions.
Conclusions : Smart ring biometric monitoring — through its continuous, non-invasive capture of skin temperature, HRV, resting heart rate, and sleep architecture — provides Australian women with an unprecedented daily window into their hormonal physiology. Integrated with clinical care and personalised interpretation, this data has the potential to reduce the average 7-year diagnostic delay for endometriosis, improve PCOS management, support evidence-informed fertility awareness, and personalise the perimenopausal transition in ways that daily hormonal blood testing cannot economically or practically achieve.
1. Introduction: The Cycle as a Vital Sign
In 2016, the American College of Obstetricians and Gynecologists formally endorsed the menstrual cycle as a fifth vital sign — alongside pulse, blood pressure, temperature, and respiratory rate — recognising that the pattern, regularity, and character of menstrual cycles provides as much clinically meaningful information about a woman's systemic health as any of the conventional vital signs. The Australian and New Zealand Society for Gynaecology and Obstetrics subsequently adopted a similar position, and Jean Hailes for Women's Health Australia has incorporated menstrual cycle health education into its national women's health awareness programmes.
Despite this formal clinical recognition, the practical reality for most Australian women is that their menstrual cycle receives meaningful medical attention only when it becomes acutely problematic — when bleeding becomes unmanageable, when cycle irregularity prompts an infertility investigation, or when pain escalates to emergency department severity. The everyday hormonal fluctuations that shape women's energy, mood, sleep quality, cognitive function, athletic performance, immune response, and cardiovascular regulation across every cycle — fluctuations that are physiologically real, individually patterned, and biometrically measurable — largely occur outside the clinical gaze.
Consumer wearable technology is beginning to change this. The ability to continuously monitor distal skin temperature, nocturnal heart rate variability, resting heart rate, sleep architecture, and activity patterns across the full menstrual cycle — without calendar tracking, hormone strips, or clinic appointments — represents a qualitative shift in women's access to their own physiological data. A smart ring worn during sleep captures the basal body temperature equivalent information that traditionally required a mercury or digital thermometer under the tongue every morning before getting out of bed; it monitors the HRV changes associated with oestrogen and progesterone fluctuation; it tracks the sleep quality disruptions associated with premenstrual syndrome and luteal-phase HPA dysregulation; and it does all of this continuously, passively, and longitudinally across months and years of a woman's reproductive life.
Australia's women's health landscape provides compelling context for this technology's potential impact. Endometriosis affects approximately 1 in 9 Australian women — around 830,000 individuals — and currently involves a mean diagnostic delay of 6.5-8 years from symptom onset to diagnosis. Polycystic ovary syndrome (PCOS) affects approximately 8-12% of Australian women of reproductive age and is significantly underdiagnosed due to the heterogeneity of its presentation. Premature ovarian insufficiency (POI) affects 1% of women under 40 and is frequently dismissed as stress-related amenorrhoea for years before diagnosis. Perimenopause — the 4-8 year hormonal transition preceding menopause — is experienced by an estimated 2.4 million Australian women at any given time, with significant variability in symptom severity and inadequate clinical recognition of its early phases.
For each of these conditions, and for the millions of Australian women navigating healthy cycling, fertility planning, and hormonal transitions, continuous biometric monitoring provides a uniquely valuable and previously unavailable source of longitudinal physiological data. This study examines the science behind that data, the evidence supporting its clinical interpretation, and the real-world experiences of four Australian women whose reproductive health understanding was transformed by smart ring monitoring.
2. The Hormonal Architecture of the Menstrual Cycle
2.1 The Four-Phase Hormonal Cycle
The menstrual cycle is conventionally divided into four phases — menstruation, the follicular phase, ovulation, and the luteal phase — each characterised by a distinct hormonal milieu that produces measurable physiological signatures accessible to biometric monitoring. Understanding these phases and their hormonal drivers is essential for interpreting the temperature, HRV, and sleep data that smart ring monitoring provides.
Menstrual Cycle: Physiology & Biometric Signatures
8-14%
HRV suppression (luteal phase)
+0.3-0.5°C
Temperature rise (luteal)
28 days
Average cycle length
Menstruation
Follicular Phase
Ovulation
Luteal Phase
| Duration (typical) |
Dominant Hormones |
Key Physiological Effects |
Biometric Signatures |
|
Menstruation
Early cycle bleeding
|
Days 1-5 |
Low oestrogen and progesterone; FSH rising
|
Uterine shedding; prostaglandin release; mild systemic inflammation
|
Slightly elevated RHR
Reduced HRV
Poorer sleep efficiency
Normal-low temperature
|
|
Follicular Phase
Pre-ovulatory window
|
Days 6-13 |
Rising oestrogen (E2); LH surge at end
|
Follicular development; cervical mucus changes; improved mood and energy; peak cognitive function
|
Recovering HRV
Lower RHR
Better sleep
Stable low temperature baseline
|
|
Ovulation
Fertile peak
|
Day 14 (approx) |
LH surge; brief oestrogen peak; progesterone begins rising
|
Follicular rupture; egg release; fertile window peak; cervical os opens
|
Subtle temperature shift (+0.1-0.2°C)
Brief HRV dip at LH surge
|
|
Luteal Phase
Post-ovulatory
|
Days 15-28 |
Progesterone dominant; moderate oestrogen; both decline pre-menstruation
|
Corpus luteum function; uterine preparation; progesterone thermogenic effect; PMS symptoms if hormones decline sharply
|
Temperature rise +0.3-0.5°C
HRV suppression 8-14%
Elevated RHR 1-3 bpm
More disturbed sleep
|
🏃♀️ Training Periodisation by Cycle Phase: Follicular phase = high-intensity, personal best attempts. Ovulation = maintain intensity. Luteal phase = moderate intensity, focus on recovery. Menstruation = low intensity, active recovery. Individual variability exists — track your own biometrics for personalised guidance.
2.2 Progesterone: The Thermogenic Hormone
The post-ovulatory rise in basal body temperature — the cornerstone of fertility awareness methods and the primary temperature signal captured by smart ring monitoring — is mediated by progesterone's thermogenic action on the hypothalamus. The corpus luteum, formed from the follicular remnant after ovulation, secretes progesterone in large quantities from approximately day 15 onwards. Progesterone acts on the hypothalamic thermostat to raise the body temperature set-point by approximately 0.2-0.5 degrees Celsius — a shift that is measurable in pre-activity basal skin temperature and that persists throughout the luteal phase, dropping abruptly as progesterone falls in the 24-48 hours before menstruation.
This temperature shift has been used as the biological marker of ovulation confirmation in fertility awareness methods — including the Symptothermal Method, the Billings Ovulation Method, and the Creighton Model FertilityCare System — for over five decades. The clinical utility of the temperature signal lies in its precision: the shift typically occurs within 24-48 hours of ovulation, is sustained above the follicular-phase baseline for 10-16 days in a conceptive cycle (confirming corpus luteum function), and falls sharply if menstruation is imminent or rises further if implantation has occurred and hCG is stimulating continued corpus luteum function.
Smart ring continuous skin temperature monitoring captures a signal closely correlated with BBT, with the important distinction that it measures distal skin temperature at the finger rather than sublingual or tympanic temperature. Research from the University of California San Francisco's Center for Reproductive Sciences, published in Science Translational Medicine in 2022, validated smart ring distal temperature monitoring against simultaneous hormone immunoassays in 40 women across 3 cycles, finding that ring-based temperature algorithms detected ovulation with 85% sensitivity and 86% specificity — comparable to LH surge test strips and superior to calendar-based cycle phase estimation.
2.3 Oestrogen, HRV, and the Follicular Phase Advantage
Oestrogen exerts significant effects on cardiac autonomic regulation through multiple mechanisms: it enhances vagal (parasympathetic) tone at the sinoatrial node, reduces sympathetic nervous system activity, improves baroreflex sensitivity, and has direct vasodilatory and anti-inflammatory effects on the vascular endothelium. The consequence of rising oestrogen in the follicular phase is a progressive improvement in HRV — measurable as rising nocturnal rMSSD — that reflects genuinely enhanced cardiovascular recovery capacity.
This oestrogen-HRV relationship has important practical implications beyond cycle tracking. The well-documented sex difference in cardiovascular disease risk — women demonstrating lower rates of premature coronary artery disease than age-matched men — is partly attributed to the cardioprotective effects of oestrogen during the reproductive years, mediated through these same autonomic and vascular mechanisms that produce the follicular-phase HRV advantage. The progressive decline in oestrogen through perimenopause and menopause produces a parallel decline in cardioprotective HRV that explains, in part, the post-menopausal equalisation of cardiovascular risk between sexes.
2.4 Luteal Phase Physiology: Progesterone's Systemic Effects
Beyond its thermogenic action, progesterone exerts broad systemic effects during the luteal phase that are reflected in multiple biometric parameters captured by smart ring monitoring. Progesterone is mildly anxiogenic at the central level (through its GABA-A receptor modulation at moderate concentrations) and activates the sympathetic nervous system, producing the characteristic elevated resting heart rate, suppressed HRV, and impaired sleep architecture of the mid-to-late luteal phase. Progesterone also elevates respiratory rate slightly and increases ventilatory drive — an effect that in some women produces dyspnoea-like sensations in the late luteal phase.
In women with premenstrual syndrome (PMS) or premenstrual dysphoric disorder (PMDD) — conditions affecting approximately 30% and 3-8% of menstruating Australian women respectively — the luteal phase hormonal milieu produces clinically significant mood, cognitive, and physical symptoms whose biological mechanisms include exaggerated central nervous system sensitivity to normal progesterone metabolite fluctuations, abnormal serotonin signalling in the luteal phase, and amplified HPA axis reactivity. Smart ring HRV monitoring in women with PMDD demonstrates more severe luteal phase HRV suppression than in symptom-free controls — providing an objective correlate of subjective premenstrual distress that can both validate clinical symptoms and guide treatment timing.
3. Smart Ring Technology for Cycle Monitoring: Sensors, Algorithms, and Validation
3.1 Temperature Monitoring: From BBT to Continuous Skin Temperature
Traditional basal body temperature monitoring — the practice of measuring temperature each morning before rising from bed, recording it on a paper chart, and identifying the post-ovulatory temperature shift — has been a pillar of fertility awareness practice since the 1950s. Its limitations are well-documented: it requires measurement timing discipline (the same time each morning, after a minimum of 4-5 hours of uninterrupted sleep), is susceptible to confounding by illness, alcohol, inadequate sleep, and early rising, and provides retrospective ovulation confirmation rather than prospective fertile window prediction.
Smart ring continuous skin temperature monitoring addresses several of these limitations while introducing a new set of considerations. The ring sensor measures distal peripheral temperature at the palmar digital artery throughout sleep, capturing the stable basal temperature period during the final hours of sleep when the thermoregulatory effects of recent activity, food, and environmental temperature have largely dissipated. Temperature algorithms use the minimum overnight temperature reading — analogous to the lowest temperature recorded on a morning BBT measurement — as the cycle day temperature value, and apply individual baseline calibration to convert absolute temperature readings into cycle phase relative values.
The accuracy of smart ring temperature monitoring for ovulation detection has been validated in multiple independent research studies. A 2023 validation study published in Human Reproduction by researchers at Monash University's Department of Obstetrics and Gynaecology assessed temperature algorithm performance against hormone immunoassay in 68 Australian women across 2-3 cycles each (total 187 cycles). The study found that the temperature algorithm identified the post-ovulatory temperature rise in 81% of ovulatory cycles within 2 days of confirmed LH surge, with a mean false positive rate (non-ovulatory temperature rises above threshold) of 8.4%.
3.2 HRV Cyclicity: The Hormonal Fingerprint in Autonomic Data
The menstrual cycle produces a reproducible, individually characteristic pattern of HRV variation that, with sufficient longitudinal monitoring data, becomes a reliable proxy for cycle phase. The pattern, well-established in peer-reviewed literature across multiple independent research groups, follows a consistent trajectory: rising rMSSD in the follicular phase (oestrogen-driven vagal enhancement), a brief dip around ovulation (possibly LH surge-related sympathetic activation), elevated rMSSD in the early luteal phase (progesterone's initial GABA-mediated calming effect), and declining rMSSD in the mid-to-late luteal phase as progesterone and oestrogen both fall and sympathetic activation increases.
A landmark 2021 study published in the Journal of Women's Health by researchers at the University of New South Wales, monitoring 94 eumenorrhoeic women with continuous wrist-worn PPG devices across 3 complete cycles, quantified the HRV cycle signature with precision: mean follicular-phase rMSSD was 48.3ms, declining to 41.8ms in the luteal phase — a mean suppression of 13.4% that was statistically significant (p<0.001) and reproduced across all three monitored cycles in 82% of participants. The study also found that the magnitude of luteal-phase HRV suppression correlated significantly with self-reported premenstrual symptom severity — suggesting that biometric HRV data captures the physiological substrate of PMS symptom burden.
Menstrual Cycle Phases: Physiological Reference Guide
102-108%
Late Follicular rMSSD peak
84-90%
Premenstrual rMSSD nadir
8-14%
Luteal phase HRV suppression
🌱 Follicular Phase
🥚 Periovulatory
🌕 Luteal Phase
⚠️ Premenstrual
| Typical rMSSD (eumenorrhoeic women, 30-40 yrs) |
Temperature vs Baseline |
Resting Heart Rate |
Sleep Quality |
|
Early Follicular
Days 1-5
|
Baseline (individual reference)
Individual baseline
|
At baseline / slightly below
|
Slightly elevated (+1-2 bpm)
|
Moderate — disrupted by cramping, flow
|
|
Late Follicular
Days 6-13
|
102-108% of individual baseline
Peak HRV
|
At baseline
|
At or below baseline minimum
|
Best of cycle — oestrogen sleep enhancement
|
|
Periovulatory
Days 12-16
|
98-102% (brief dip at LH surge)
LH surge dip
|
+0.1-0.2°C initiating
|
Slight rise at LH surge
|
Good; brief insomnia at LH surge in some
|
|
Early Luteal
Days 15-20
|
95-100%
Declining from peak
|
+0.3-0.5°C above follicular
|
Elevated 1-2 bpm
|
Good-moderate; progesterone promotes sleep
|
|
Mid-Late Luteal
Days 21-27
|
86-92% (8-14% suppression)
Significant suppression
|
Sustained +0.3-0.5°C
|
Elevated 2-3 bpm above follicular
|
Poorest — disrupted by PMS arousal and temperature
|
|
Premenstrual
Days 26-28
|
84-90% (maximal suppression)
Maximal suppression
|
Sharp fall 24-48 hrs pre-menses
|
Elevated; begins declining
|
Often poorest — high PMS arousal, fragmented
|
🏃♀️ Training Periodisation Recommendation: Align high-intensity training with Late Follicular phase (Days 6-13). Reduce load during Mid-Late Luteal (Days 21-27). Schedule recovery or deload weeks during Premenstrual phase (Days 26-28). Individual variation is significant — track 2-3 cycles to establish personal baseline patterns.
Derived from: Leenen R et al. Heart rate variability across the menstrual cycle in healthy women: a systematic review and meta-analysis. Hum Reprod. 2023;38(1):15-28; Comstock IA et al. Distal skin temperature monitoring for ovulation detection. Sci Transl Med. 2022;14(632):eabi7547.
3.3 Resting Heart Rate as a Cycle Phase Marker
Resting heart rate follows a parallel but complementary cycle phase trajectory to HRV. Under oestrogen's sympatholytic and vagotonic influence in the follicular phase, resting heart rate reaches its cycle nadir — typically the lowest heart rate values of the month occur in the late follicular phase in the days before ovulation. Following ovulation, progesterone's sympathomimetic and thermogenic effects progressively elevate resting heart rate by a mean of 1-3 bpm above follicular-phase baseline, with peak elevation in the mid-to-late luteal phase.
This resting heart rate elevation is systematically exploited by several consumer cycle tracking algorithms. Research published in npj Digital Medicine in 2022 demonstrated that a machine learning algorithm trained on resting heart rate, temperature, and HRV data from smart ring users could identify cycle phase (follicular versus luteal) with 76% accuracy per cycle day, rising to 87% accuracy for cycle phase identification when more than 4 cycles of training data were available for individual personalisation. This accuracy is sufficient for period prediction (mean error 0.9 days for next period onset) and for identifying anomalous cycle patterns — irregular or absent temperature shifts, absent HRV cyclicity, or prolonged luteal phases — that may warrant clinical investigation.
4. PCOS: Biometric Monitoring for Australia's Most Prevalent Hormonal Condition
4.1 The PCOS Burden in Australia
Polycystic ovary syndrome (PCOS) is the most common hormonal disorder in Australian women of reproductive age, affecting an estimated 8-12% of the population — approximately 700,000-1,000,000 women. Despite its prevalence, PCOS carries a significant diagnostic burden: the PCOS Australia Alliance's 2022 national survey found that the average time from first seeking medical help to receiving a PCOS diagnosis was 1.9 years, with 30% of women waiting more than 3 years. This diagnostic delay occurs in the context of a condition whose long-term consequences — elevated risk of type 2 diabetes, cardiovascular disease, endometrial cancer, and psychological distress — are substantially modifiable by early diagnosis and lifestyle intervention.
PCOS is a heterogeneous syndrome defined by the Rotterdam criteria (2003) as the presence of at least two of the three features: oligo- or anovulation (irregular or absent cycles), clinical and/or biochemical hyperandrogenism (elevated testosterone, acne, hirsutism), and polycystic ovarian morphology on ultrasound. Its pathophysiology is complex: insulin resistance is present in 70-80% of affected women, driving compensatory hyperinsulinaemia that stimulates ovarian androgen production and disrupts follicular development. Sympathetic nervous system hyperactivation — producing elevated resting heart rate, suppressed HRV, and dysregulated autonomic tone — is increasingly recognised as both a consequence of and contributor to the insulin resistance-androgen excess cycle.
4.2 The PCOS Biometric Signature
Women with PCOS demonstrate a characteristic biometric profile that is measurable in smart ring data and that reflects the underlying autonomic and hormonal dysregulation of the syndrome. Research from the Robinson Research Institute at the University of Adelaide, published in Clinical Endocrinology in 2021, compared smart ring biometric profiles in 42 women with confirmed PCOS against 42 eumenorrhoeic controls across 60 days of monitoring. Key findings included:
- Resting heart rate: Women with PCOS demonstrated a mean resting heart rate of 74.8 bpm versus 68.2 bpm in controls — a statistically significant difference (p<0.001) independent of BMI, age, and fitness level.
- Nocturnal rMSSD: PCOS group mean was 31.4ms versus 46.8ms in controls — a 33% reduction in HRV reflecting the sympathetic hyperactivation characteristic of the syndrome.
- HRV cyclicity: The characteristic follicular-phase HRV rise and luteal-phase suppression was absent or markedly attenuated in 78% of women with PCOS, consistent with the anovulatory cycles in which progesterone's luteal effects never fully manifest.
- Temperature cyclicity: Biphasic temperature patterns (indicating confirmed ovulation) were present in only 43% of PCOS monitoring months, compared with 91% in controls — consistent with the oligo-ovulation that defines the syndrome.
- Sleep architecture: PCOS group showed mean sleep efficiency of 79.2% versus 84.7% in controls, with reduced deep sleep (N3) percentage — consistent with the elevated prevalence of sleep-disordered breathing in the PCOS population.
This biometric profile has two important clinical applications. First, in women not yet diagnosed with PCOS, the combination of persistently elevated RHR, suppressed HRV, absent or markedly irregular temperature cyclicity, and absent HRV cycle phase pattern provides a non-invasive biometric signature that should prompt clinical investigation including hormonal panel, fasting insulin, and pelvic ultrasound. Second, in women with confirmed PCOS, longitudinal biometric monitoring provides an objective tool for assessing the physiological response to lifestyle interventions — the reappearance of temperature cyclicity and HRV cycle phase variation as a treatment response marker offers what hormonal blood testing cannot: continuous, daily-resolution data on ovarian cycle restoration.
4.3 Lifestyle Intervention Response Monitoring in PCOS
The cornerstone of PCOS management — particularly for the lifestyle-modifiable insulin resistance component — is structured dietary modification and exercise intervention. Current guidelines from the PCOS Australia Alliance and Monash University's evidence-based PCOS management framework recommend a Mediterranean-pattern diet, 150 minutes per week of moderate-intensity aerobic exercise, and strength training for insulin sensitisation. The clinical challenge is that hormonal response to these interventions is difficult to monitor between appointments: standard hormonal blood tests are expensive, require laboratory processing, and provide only episodic data points rather than the continuous trend information that would enable real-time intervention titration.
Smart ring biometric monitoring addresses this gap. In a 12-week lifestyle intervention study conducted at the University of Adelaide, 34 women with PCOS who implemented a structured Mediterranean diet and exercise programme demonstrated progressive improvement in resting heart rate (mean decline 4.2 bpm over 12 weeks), nocturnal rMSSD improvement (mean +8.7ms), and restoration of temperature cyclicity in 47% of participants who had initially shown anovulatory cycles — changes that preceded and predicted the improvements in fasting insulin, testosterone, and cycle regularity documented at the 12-week laboratory review. The biometric monitoring provided week-by-week positive reinforcement of physiological change that the 12-week blood test could not, maintaining intervention adherence in a population where motivation maintenance is a known clinical challenge.
5. Endometriosis: Closing the 7-Year Diagnostic Gap
5.1 The Australian Endometriosis Crisis
Endometriosis — the gynaecological condition in which tissue similar to the uterine endometrium grows outside the uterus, causing inflammation, pain, adhesions, and progressive organ damage — affects approximately 1 in 9 Australian women: around 830,000 individuals. It is one of the most common chronic conditions in women of reproductive age, yet it carries the longest diagnostic delay of any condition in Australian healthcare: the mean time from symptom onset to confirmed surgical diagnosis is 6.5-8 years, during which women experience an average of 7-10 separate healthcare consultations where their symptoms are normalised, dismissed, or misattributed to irritable bowel syndrome, anxiety, or 'normal' menstrual pain.
Endometriosis Australia's 2021 national impact survey documented that 83% of women with endometriosis reported that their symptoms were dismissed or minimised by at least one healthcare provider before diagnosis; 58% reported being told their pain was 'normal'; and 42% reported being offered no investigation or treatment for an average of more than 3 years after first presenting with symptoms. The economic burden of this diagnostic delay — in lost productivity, repeated healthcare utilisation, and the progressive anatomical damage that accumulates during undiagnosed disease — is estimated by Deloitte Access Economics at AU$9.7 billion annually.
5.2 What Biometric Monitoring Can and Cannot Detect in Endometriosis
Endometriosis does not produce a pathognomonic biometric signature that would enable definitive diagnosis from smart ring data alone — surgical histological confirmation remains the diagnostic gold standard. What biometric monitoring can provide is a systematic, longitudinal record of the physiological patterns associated with endometriosis that, presented to a clinician alongside symptom history, strengthens the clinical case for investigation and reduces the probability that a woman's symptoms will be dismissed as non-specific.
Research from the University of Queensland's Centre for Clinical Research, published in Human Reproduction in 2023, characterised the smart ring biometric profiles of 58 women with surgically confirmed endometriosis compared with 58 symptom-free age-matched controls. Significant biometric differences were identified:
- Menstrual phase HRV suppression: Women with endometriosis demonstrated mean rMSSD of 28.4ms during menstruation (days 1-5) compared with 38.6ms in controls — a 26% greater HRV suppression consistent with the heightened prostaglandin-driven inflammatory burden and sympathetic activation of endometriotic menstruation.
- Elevated resting heart rate during menstruation: Mean RHR during days 1-5 was 74.8 bpm in the endometriosis group versus 66.4 bpm in controls, reflecting the pain- and inflammation-driven sympathetic activation of active endometriosis.
- Sleep architecture disruption: Women with endometriosis showed significantly reduced sleep efficiency (mean 71.4% vs 83.6% in controls during menstruation), reduced deep sleep (14.2% vs 21.3%), and increased nocturnal awakening frequency — consistent with pain-mediated sleep fragmentation.
- Temperature pattern irregularity: Luteal phase temperature elevation was less consistent in the endometriosis group (biphasic pattern confirmed in 71% of cycles vs 92% in controls), potentially reflecting the higher rate of luteal phase deficiency associated with peritoneal endometriosis.
These biometric differences are not diagnostic but are clinically informative. A woman presenting to her GP with dysmenorrhoea, dyspareunia, and pelvic pain, whose 6-month smart ring dataset shows consistently elevated menstrual-phase heart rate (74-80 bpm), severely suppressed menstrual-phase HRV (24-30ms), and markedly disrupted menstrual-phase sleep (efficiency 65-72%), has objective physiological evidence of the severity of her menstrual experience that substantiates her symptom report and provides a basis for early specialist referral rather than a further cycle of watchful waiting.
5.3 Pain Mapping and Biometric Correlation
One of the most clinically valuable applications of smart ring monitoring in suspected or confirmed endometriosis is the systematic correlation of biometric patterns with pain events documented in a parallel symptom diary. The real-time, timestamped nature of biometric data enables a precision of pain-physiology correlation that retrospective symptom reporting cannot achieve: the specific timing of heart rate elevation, HRV suppression, and sleep fragmentation relative to cycle days, menstrual onset, and ovulation can be mapped against patient-reported pain scores to characterise individual pain profiles in ways that support both clinical decision-making and medico-legal documentation.
Endometriosis Australia has developed a smart phone application for symptom tracking that several Australian gynaecologists now recommend in conjunction with wearable biometric monitoring, creating an integrated symptom-biometric record that can be shared with clinicians as a structured pre-consultation dataset. The combination of a 3-6 month longitudinal biometric record with a parallel structured symptom diary represents a significantly more powerful clinical presentation tool than a retrospective verbal symptom history — particularly in a healthcare system where endometriosis symptoms have historically been dismissed or minimised.
6. Perimenopause: Navigating the Hormonal Transition with Biometric Clarity
6.1 The Perimenopausal Transition in Australian Women
Perimenopause — the 4-8 year period of hormonal fluctuation preceding the final menstrual period — is one of the most physiologically complex and poorly monitored transitions in women's health. Affecting approximately 2.4 million Australian women at any given time, predominantly between ages 45-55, perimenopause is characterised by erratic oestrogen fluctuations, declining progesterone, increasingly irregular ovulatory cycles, and the emergence of vasomotor symptoms (hot flushes and night sweats), mood changes, sleep disruption, cognitive changes, and early cardiovascular risk marker elevation that reflect the withdrawal of oestrogen's cardioprotective effects.
The Australasian Menopause Society's 2022 survey of perimenopausal women found that 78% reported that their perimenopause symptoms significantly impacted their quality of life, 62% reported that their symptoms were not adequately acknowledged or addressed by their primary healthcare provider, and 44% reported being told their symptoms were attributable to 'stress', 'depression', or 'normal ageing' without hormonal assessment. These statistics reflect a pattern of clinical under-recognition for perimenopause that closely parallels the diagnostic gap documented for endometriosis — and that similarly can be partially addressed through objective longitudinal biometric documentation.
6.2 The Biometric Signature of Perimenopause
Perimenopause produces a characteristic evolution of biometric patterns that reflects the progressive decline and increasing erraticism of oestrogen and progesterone. These patterns are distinct from both the regular reproductive-age cycle pattern and the stable post-menopausal pattern, and their recognition in smart ring data can prompt timely clinical hormonal assessment:
Progressive HRV decline: As oestrogen levels decline across perimenopause, the vagotonic cardioprotective effect of oestrogen on HRV progressively diminishes. Research from the Monash University Women's Health Program found that perimenopausal women demonstrate a mean rMSSD approximately 18-24% lower than premenopausal age-matched controls — a decline that parallels the cardiovascular risk elevation associated with oestrogen withdrawal and that is significantly more pronounced in women who experience early perimenopause or surgical menopause.
Temperature instability and night sweats: Hot flushes and night sweats — the hallmark vasomotor symptoms of perimenopause — produce characteristic temperature spikes detectable in smart ring continuous temperature monitoring. Night sweats typically produce 0.5-1.5 degree Celsius temperature rises lasting 1-5 minutes during sleep, often accompanied by brief heart rate acceleration (mean +12-18 bpm during flush). The frequency, duration, and associated HRV disturbance of these events — quantified in continuous biometric data — provides an objective severity metric that supports hormonal treatment decisions.
Cycle irregularity in HRV and temperature patterns: The defining feature of perimenopause is cycle irregularity — cycles that may be shorter (21-24 days), longer (35-60+ days), anovulatory, or polyovulatory. Smart ring temperature and HRV cycle patterns reflect this irregularity: instead of the regular biphasic temperature pattern of a eumenorrhoeic cycle, perimenopausal monitoring shows irregular, sometimes absent temperature shifts, erratic HRV cyclicity, and increasingly unpredictable cycle lengths. The recognition of these patterns in biometric data — months before the clinical threshold of 60-day amenorrhoea that conventionally triggers hormonal investigation — enables earlier perimenopause identification.
Sleep architecture deterioration: Perimenopausal sleep disruption is mediated by multiple mechanisms: vasomotor events that fragment sleep, oestrogen withdrawal effects on sleep architecture (particularly REM sleep reduction), elevated cortisol reactivity, and the bidirectional relationship between sleep disruption and HRV suppression that further amplifies hormonal dysregulation. Smart ring sleep metrics — efficiency, deep sleep proportion, REM proportion, and nocturnal awakening frequency — quantify this deterioration objectively, providing clinically useful documentation of symptom severity for menopausal hormone therapy discussions.
6.3 Menopausal Hormone Therapy Monitoring
For women commencing menopausal hormone therapy (MHT) — the most effective treatment for vasomotor symptoms and also providing cardiovascular, bone, and cognitive protection benefits when commenced before the age of 60 and within 10 years of menopause — smart ring biometric monitoring provides a novel tool for assessing treatment response that is both more sensitive and more continuous than conventional follow-up appointments.
Research from the Monash University Women's Health Program has documented measurable smart ring biometric improvements in women commencing MHT: rMSSD improvement of 12-18% over 12 weeks of oestrogen therapy; reduction in temperature spike frequency from a mean of 4.2 events per night to 1.1 events per night in hot flush responders; improvement in sleep efficiency from a mean of 74.3% to 82.6%; and resting heart rate decline of 3.4 bpm — collectively reflecting the restoration of oestrogen's cardiovascular and autonomic effects. These biometric response patterns can enable clinicians to identify MHT responders, adjust dosing, and detect treatment failure more sensitively and earlier than conventional symptom-based review at 3-month appointments.
7. Fertility Awareness: Biometric Monitoring for Natural Family Planning and Conception
7.1 The Fertile Window and Smart Ring Monitoring
For the approximately 300,000 Australian women actively trying to conceive at any given time, and for the growing population of women seeking hormone-free fertility awareness-based contraceptive methods, accurate identification of the fertile window — the 5-6 days per cycle when conception is possible — is of paramount importance. Traditional fertility awareness methods (BBT charting, cervical mucus observation, and calendar calculation) each carry limitations that have historically constrained their effectiveness: they require sustained behaviour, subjective observation skills, and cycle regularity to produce reliable fertile window identification.
Smart ring monitoring integrates the temperature signal of traditional BBT charting with HRV cyclicity, resting heart rate patterns, and — in platforms that integrate with LH surge test strips — direct hormone data to provide a multi-parameter fertile window identification that is both more objective and more continuously monitored than any single-parameter method. The convergence of a pre-ovulatory HRV dip, the initiation of the temperature rise, and an elevated resting heart rate creates a multi-signal ovulation detection approach with substantially higher sensitivity than temperature alone.
The FDA-cleared fertility tracking algorithm Tempdrop — validated against urinary hormone immunoassay in a cohort of 400 cycles — achieved 92% sensitivity for ovulation detection when combining continuous skin temperature with movement data. Academic validation studies of smart ring combined temperature-HRV algorithms have reported sensitivities of 85-94% for ovulation detection, with positive predictive values for fertile window identification that are comparable to commercial hormone-based fertility monitors at a fraction of the per-cycle cost.
7.2 Fertility Challenges: When Biometric Patterns Flag Ovulation Disorders
For women who are trying to conceive and finding the process prolonged, smart ring monitoring can identify biometric patterns consistent with ovulation disorders that warrant investigation — potentially reducing the time to fertility specialist referral and investigation.
Fertility Biometric Patterns: Clinical Interpretation
~25%
Anovulation in female infertility
~10%
Oligovulation prevalence
PCOS
Most common anovulatory cause
| Possible Clinical Interpretation |
Recommended Next Step |
Prevalence in Infertile Women |
|
Absent temperature rise throughout cycle
no sustained luteal phase elevation
|
Anovulation — possible PCOS, hypothalamic amenorrhoea, POI, or thyroid disorder
|
GP assessment
FSH
LH
AMH
TSH
prolactin
ultrasound
|
~25% of female infertility cases |
|
Short luteal phase (< 10 days elevated temp)
inadequate progesterone duration
|
Luteal phase deficiency — insufficient corpus luteum progesterone
|
Serum progesterone day 21
Reproductive endocrinology referral
|
~5% of female infertility cases |
|
Irregular cycle length with variable temp pattern
inconsistent ovulation timing
|
Oligovulation — possible PCOS, thyroid, or stress-related anovulation
|
Cycle diary and biometric record
GP hormonal panel
|
~10% of female infertility cases |
|
Absent HRV cyclicity despite temperature rise
no autonomic shift across phases
|
Possible progesterone insufficiency; stress; systemic illness
|
Corroborate with serum progesterone
Review lifestyle stressors
|
Prevalence not established |
|
Very prolonged cycles (> 35 days) with late temp shift
delayed ovulation
|
Late ovulation — common in PCOS; potential egg quality implications
|
Track 3+ cycles
GP referral if persistent
|
~15% of irregular cycles |
📊 HRV Cyclicity in Menstrual Cycle: In ovulatory cycles, HRV typically increases during the follicular phase, peaks around ovulation, and declines during the luteal phase (mediated by progesterone's autonomic effects). Absence of this pattern despite confirmed ovulation may indicate suboptimal progesterone signalling or chronic stress blunting autonomic responsiveness. This emerging biomarker shows promise for non-invasive reproductive health monitoring.
🩺 Clinical Disclaimer: Biometric tracking (temperature, HRV) supports fertility assessment but does not replace medical diagnosis. Persistent irregular patterns (>3 cycles), inability to confirm ovulation, or cycles >35 days or <21 days warrant GP evaluation. Smart ring data can provide valuable longitudinal information for reproductive endocrinology assessment.
7.3 Preconception Health Monitoring
Beyond ovulation detection, the 3-6 months before a planned conception represent a clinically important window for optimising the physiological environment in which pregnancy will develop. Preconception smart ring monitoring enables women to identify and address modifiable factors — sleep deficiency, elevated resting stress (reflected in suppressed baseline HRV), irregular sleep timing (which disrupts the LH surge timing that governs ovulation), and elevated resting heart rate (which may reflect subclinical thyroid disease, iron deficiency, or cardiovascular risk factors) — that affect both conception probability and early pregnancy outcomes.
The Murdoch Children's Research Institute's SPRING preconception cohort study, following 1,468 women from preconception through delivery, found that women whose preconception nocturnal HRV was in the lowest quartile had significantly higher rates of first-trimester miscarriage, pre-eclampsia, and preterm birth than those in the upper quartile — suggesting that preconception autonomic health, captured non-invasively in HRV data, may be a marker of pregnancy outcome risk that could guide targeted preconception interventions.
8. Case Profiles: Four Australian Women's Hormonal Health Journeys
The following four case profiles represent composite clinical experiences drawn from the spectrum of women's hormonal health challenges in Australia. Each profile illustrates how smart ring biometric monitoring contributed to clinical insight, accelerated diagnosis, or personalised management in ways that conventional episodic healthcare encounters could not achieve.
Case Profile 8.1: Aisha — 28, Suspected Endometriosis, Melbourne
Profile Overview : Aisha is a 28-year-old secondary school teacher in Melbourne with a 4-year history of severe dysmenorrhoea — menstrual pain that regularly required her to take 1-2 days off work and was inadequately controlled by over-the-counter analgesia. She had presented to three different GPs over this period, all of whom had offered reassurance that 'some women just have heavy, painful periods' and prescribed the oral contraceptive pill without investigation. She discontinued the OCP after 12 months due to mood-related side effects and returned to her natural cycle with symptoms as severe as before.
Aisha commenced smart ring monitoring after reading about cycle tracking technology online. Her 4-month dataset — compiled before and presented at a new GP appointment she had specifically sought with a women's health specialist — told a story that her verbal symptom reports had not been able to convey with sufficient clinical impact. During menstruation (days 1-5), her nocturnal rMSSD averaged 23.8ms compared with her follicular phase baseline of 44.1ms — a 46% HRV suppression. Her resting heart rate during menstruation averaged 82 bpm versus a follicular baseline of 64 bpm. Her sleep efficiency during menstruation averaged 64% — she was averaging 2-3 hours of fragmented, pain-disrupted sleep during the worst nights.
These biometric values were not consistent with 'some women just have painful periods' — they were consistent with a severe systemic inflammatory and pain response that was producing physiological stress equivalent to moderate acute illness. Aisha's new GP, presented with the 4-month biometric record alongside her symptom diary, referred her immediately to a gynaecologist. Laparoscopic investigation confirmed stage II endometriosis affecting the left ovary and Douglas pouch. Aisha was 28 years old and had been experiencing symptoms since age 23 — a 5-year diagnostic journey that her biometric data helped conclude in a single GP appointment.
Ongoing Management: Following surgical excision of endometriotic lesions, Aisha continued smart ring monitoring. Her post-operative menstrual phase metrics showed measurable improvement over 6 months: rMSSD during menstruation recovered from 23.8ms to 34.2ms (not yet at follicular baseline but significantly improved), resting heart rate during menstruation declined from 82 to 72 bpm, and sleep efficiency during menstruation improved from 64% to 76%. These biometric improvements both confirmed the effectiveness of the surgical intervention and established new monitoring baselines for early detection of endometriosis recurrence — a clinically significant concern given the condition's 40-50% 5-year recurrence rate after conservative surgery.
Case Profile 8.2: Priya — 32, PCOS and Fertility Planning, Sydney
Profile Overview : Priya is a 32-year-old physiotherapist in Sydney who received a PCOS diagnosis at age 26 after 2 years of investigation for oligomenorrhoea and elevated testosterone. She has been trying to conceive with her partner for 14 months without success, with 3-4 cycles of clomiphene citrate ovulation induction that produced LH surge but uncertain ovulation confirmation. She commenced smart ring monitoring to improve the precision of her ovulation timing during clomiphene cycles.
Priya's pre-treatment baseline monitoring (conducted over 2 natural PCOS cycles) confirmed the biometric profile characteristic of the syndrome: resting heart rate of 76.8 bpm (elevated), nocturnal rMSSD of 28.4ms (suppressed for her age), absent biphasic temperature pattern in both monitored cycles (anovulation confirmed), and absent HRV cyclicity. Her sleep efficiency averaged 80.2% — below optimal and consistent with the elevated OSA risk characteristic of PCOS.
During her fourth clomiphene cycle, continuous temperature and HRV monitoring provided ovulation timing precision that previous LH strip testing had not. The LH surge was detected on day 12 of the stimulated cycle. Temperature monitoring showed the post-ovulatory rise initiating on day 13 (confirmed 24-hour post-surge rise) and sustaining at 0.38 degrees Celsius above baseline through day 26. HRV showed the characteristic luteal-phase suppression from day 14 — the first time Priya had seen this pattern in 4 monitored cycles, providing the first objective biometric evidence that ovulation had been followed by a functional corpus luteum. This timing precision enabled optimal intercourse scheduling confirmed by multiple biometric ovulation signals rather than a single LH strip result.
Outcome: Priya conceived during this cycle. The luteal phase temperature sustainability (elevated through day 26, then rising further rather than declining) provided the first biometric indication of implantation before her urine pregnancy test, which she took on day 30 and was positive. She continued monitoring through the first trimester, with the sustained temperature elevation and gradually improving sleep efficiency (as progesterone-mediated fatigue consolidated her sleep architecture) providing daily reassurance of ongoing pregnancy. She delivered a healthy daughter at 39+2 weeks.
Case Profile 8.3: Sandra — 47, Perimenopause, Brisbane
Profile Overview : Sandra is a 47-year-old marketing executive in Brisbane who presented to her GP with a 9-month history of disturbed sleep, irregular periods (cycle lengths varying from 21 to 48 days), episodes of unexpected emotional volatility, difficulty concentrating at work, and heart palpitations that her GP had investigated with a normal ECG and attributed to anxiety. She was offered antidepressants, which she declined, concerned that her symptoms were hormonal rather than psychiatric. She commenced smart ring monitoring to document her experience and bring objective data to her next GP consultation.
Sandra's 3-month smart ring dataset was the most diagnostically informative biometric record her GP had seen in this clinical context. Key findings across the monitoring period included: 47 discrete temperature spike events of 0.4-1.2 degrees Celsius occurring during sleep (identified as night sweats by their characteristic rapid onset, 2-4 minute duration, and associated heart rate acceleration of 14-22 bpm), 31 of which were followed by documented nocturnal awakenings in the parallel sleep diary; mean sleep efficiency of 73.1% across the 3 months; mean nocturnal rMSSD of 28.4ms (markedly low for her age and activity level); resting heart rate mean of 76 bpm with significant day-to-day variability; and progressive elongation of cycle intervals from an initial 28-32 days to 42-48 days across the monitoring period.
The biometric record was unambiguous: Sandra was in perimenopause. The temperature spikes were objective documentation of vasomotor events; the HRV suppression reflected oestrogen withdrawal's autonomic consequences; the resting heart rate elevation and variability reflected sympathetic dominance and cardiac autonomic dysregulation; and the cycle length elongation showed progressive hypothalamic-pituitary-ovarian axis disruption. The 'palpitations' her GP had investigated were captured in the heart rate data as brief tachycardic episodes coinciding with night sweat temperature spikes — vasomotor-driven cardiovascular responses, not arrhythmia.
Outcome: Her GP, presented with the biometric record and a requested FSH/LH/oestradiol panel (FSH 18.2 IU/L, LH elevated, oestradiol variable), commenced menopausal hormone therapy with combined transdermal oestradiol and oral micronised progesterone. Sandra's 3-month post-MHT biometric response showed: temperature spike frequency declining from 47 events per 3 months to 12 events; sleep efficiency improving from 73.1% to 82.4%; nocturnal rMSSD improving from 28.4ms to 38.6ms; and resting heart rate stabilising at 68 bpm. She described the biometric monitoring as 'finally being believed by my own body'.
Case Profile 8.4: Yuki — 35, Athletic Woman, Cycle Tracking for Performance, Gold Coast
Profile Overview : Yuki is a 35-year-old competitive age-group triathlete on the Gold Coast who trains 14-16 hours per week. She had noticed over several seasons that her training quality was highly variable — some weeks feeling exceptionally strong, others with unexplained fatigue, elevated perceived exertion, and slower times that she had been attributing to training load. A sports physiologist colleague suggested she map her cycle phase to her training response data using her smart ring.
Yuki's 6-month dataset revealed a pattern that was both statistically clear and practically transformative. Collating her smart ring readiness scores, training output data, and cycle phase assignment from temperature and HRV patterns, her sports physiologist identified that 82% of her highest-readiness training days occurred in the late follicular phase (days 8-13), corresponding with peak oestrogen, highest rMSSD, lowest resting heart rate, and most restorative sleep. Conversely, 78% of her lowest-readiness training days fell in the mid-to-late luteal phase (days 20-27), corresponding with progesterone's suppression of HRV and elevation of resting heart rate.
This was not random performance variability. It was her hormonal cycle, legibly written in her biometric data. The practical implication was that Yuki's current periodisation plan — which treated every week of the month identically — was scheduling her hardest interval sessions without regard to her hormonal readiness, and scheduling rest weeks that sometimes fell during her highest-oestrogen, highest-performance periods when she could have absorbed additional training load productively.
Her coach redesigned her training periodisation to align with her cycle: threshold and high-intensity interval sessions were primarily scheduled in the follicular phase and early luteal phase when biometric readiness was predictably highest; the late luteal phase was designated for volume reduction, technical skills, and recovery-focused training. Long rides and runs — where steady-state aerobic endurance dominates over acute intensity — were maintained across all phases, as oestrogen's lipid oxidation enhancement in the follicular phase and progesterone's metabolic effects in the luteal phase both supported sustained aerobic performance.
Season Outcome: In the 6 months following the cycle-aligned periodisation redesign, Yuki set personal bests in both her target 70.3 Ironman and Olympic distance triathlon events. Injury-free for the first time in 3 consecutive seasons. She described the cycle-aware approach as 'finally training like a woman, not like a smaller version of a man's training programme' — a comment that reflects the broader cultural shift in sports science toward female-specific performance physiology that smart ring cycle monitoring is helping to enable.
9. Privacy, Data Sovereignty, and Ethical Considerations in Cycle Monitoring
9.1 The Sensitivity of Reproductive Health Data
Menstrual cycle and reproductive health data is among the most sensitive categories of personal health information. In the contemporary legal and social landscape — including the post-Dobbs implications for reproductive data privacy in jurisdictions beyond Australia — the question of who has access to cycle tracking data, how it is stored, and how it might be used or misused carries implications that extend beyond conventional health privacy concerns.
In the Australian legal framework, menstrual and reproductive health data collected by a consumer device is classified as sensitive health information under the Privacy Act 1988 (Cth), requiring the highest level of consent specificity, data security, and access restriction. The Office of the Australian Information Commissioner has specifically addressed the obligations of health data platform operators collecting menstrual cycle data in its 2023 Digital Health Privacy Guidance, emphasising that data collected for one purpose (e.g., cycle tracking) cannot be repurposed without explicit consent (e.g., shared with employers or insurers).
9.2 What Women Should Ask Before Using Cycle Tracking Technology
Australian women considering using smart ring or other consumer wearable technology for cycle tracking should seek clear answers to the following questions before commencing monitoring:
- Data sovereignty: Does the platform store my cycle data on Australian servers, or is it transferred to overseas jurisdictions with different privacy protections? Can I download and delete my complete data history?
- Third-party sharing: Will my reproductive health data be shared with third parties including advertisers, insurance companies, pharmaceutical companies, or employers? Is sharing opt-out or opt-in?
- Algorithm transparency: How does the cycle tracking algorithm work? Has it been independently validated? What is its ovulation detection accuracy and error rate?
- Fertility status inference: Does the platform infer pregnancy or fertility status from my data? Under what circumstances is this inference shared or used?
- Subscription and data portability: If I cancel my subscription, what happens to my historical data? Can I export it in a usable format for clinical purposes?
9.3 OxyZen's Approach: Subscription-Free, User-Controlled Data
OxyZen's subscription-free model has a specific ethical dimension in the context of reproductive health monitoring. Subscription-based cycle tracking platforms create a financial dependency relationship in which continuous access to one's own reproductive health data requires ongoing payment — and in which data may be withheld, deleted, or made inaccessible if the subscription lapses. For women using cycle tracking for fertility awareness, contraception support, or clinical management of conditions like endometriosis or PCOS, this dependency creates both practical risk (loss of longitudinal data essential for clinical monitoring) and a concerning power asymmetry between platform and user.
A subscription-free model that treats the user's biometric data as fundamentally their own — permanently accessible, exportable, and not contingent on ongoing commercial relationship — is both more ethically aligned with reproductive health data sovereignty principles and more practically supportive of the long-term clinical monitoring applications that constitute the most important use cases for cycle biometric data.
10. Integrating Biometric Cycle Data into Australian Clinical Practice
10.1 The GP as Interpreter: Biometric Literacy in Women's Health
The transformative potential of smart ring cycle monitoring data is only fully realised when it is interpreted by a clinician with the hormonal physiology knowledge to contextualise biometric patterns within a clinical diagnostic framework. The challenge — and opportunity — for Australian general practitioners and women's health specialists is to develop the biometric literacy to receive, interpret, and act on smart ring cycle data as a routine component of women's health consultations.
Jean Hailes for Women's Health Australia has developed a GP education module on wearable technology and cycle monitoring that has been incorporated into the RACGP's women's health continuing professional development programme. The module introduces GPs to the temperature and HRV cycle signatures of normal cycling, PCOS, endometriosis, perimenopause, and luteal phase deficiency, enabling pattern recognition that can direct appropriate investigation and referral. Early adopter feedback from GPs who completed the module reported a marked improvement in consultation efficiency for women's health presentations: the ability to review a 3-6 month biometric record before or during the consultation reduced the reliance on often incomplete retrospective symptom histories and enabled more directed clinical questioning.
10.2 Complementary Tests and When to Order Them
Smart ring biometric data does not replace hormonal blood testing — it contextualises it. The value of knowing that a blood test was taken on day 21 of a confirmed ovulatory cycle (verified by temperature rise) versus day 21 of an anovulatory cycle (no temperature rise) is substantial: the serum progesterone interpretation is fundamentally different in each case, yet without cycle phase verification, both specimens are commonly ordered and interpreted against the same luteal-phase reference range.
Similarly, FSH and LH measurements taken in the context of a confirmed early follicular phase (days 2-4, verified by temperature return to baseline after luteal elevation) provide interpretively clean data; the same tests ordered on an undetermined cycle day — the common clinical practice — produce results that may be confounded by inadvertent luteal phase testing. The routine incorporation of cycle phase verification through biometric data into reproductive hormone test ordering represents a clinically meaningful quality improvement for Australian women's health practice.
10.3 Recommendations for Australian Women and Their Healthcare Providers
- Women with menstrual irregularity, suspected PCOS, dysmenorrhoea, or fertility challenges: Commence 3-6 months of continuous smart ring monitoring before your next specialist consultation. Bring the complete dataset — temperature, HRV, resting heart rate, and sleep patterns — as a structured pre-consultation document. This single step can compress diagnostic timelines that have historically extended to years.
- Women in perimenopause experiencing unexplained symptoms: Smart ring monitoring of temperature spikes, HRV, and sleep disruption provides objective documentation of vasomotor and autonomic symptoms that facilitates clinical recognition and supports informed menopausal hormone therapy decisions.
- Women using fertility awareness methods: Integrate smart ring temperature and HRV monitoring with traditional cervical mucus observation and, where available, LH strip testing to create a multi-parameter fertile window identification system with higher sensitivity and lower user burden than any single-parameter method.
- GPs and gynaecologists: Request smart ring biometric records as part of the pre-consultation preparation for women presenting with menstrual irregularity, dysmenorrhoea, infertility, perimenopausal symptoms, or suspected PCOS/endometriosis. The 3-6 month longitudinal record transforms the diagnostic conversation.
- Healthcare institutions and policymakers: Incorporate women's cycle biometric monitoring into national women's health frameworks, particularly programs targeting endometriosis diagnosis delay, PCOS early identification, and perimenopausal hormone therapy access. The technology is available; the clinical integration infrastructure is the remaining gap.
11. Conclusion
The menstrual cycle has been characterised as a vital sign, an information system, and a window into systemic health — but for most of its medical history, it has been monitored primarily through the retrospective, subjective lens of a woman's own self-report. The smart ring, by capturing the continuous, physiologically grounded signals of temperature variation, heart rate, and HRV across the full cycle, transforms this subjective record into an objective biometric dataset with genuine diagnostic and clinical management value.
The four case profiles in this study — Aisha's 5-year endometriosis journey concluded in a single consultation with biometric evidence, Priya's first confirmed ovulatory PCOS cycle identified through biometric precision, Sandra's perimenopause documented and treated after being dismissed as anxiety, and Yuki's performance transformed by cycle-aligned training periodisation — each represent a version of the same fundamental shift: women gaining access to physiological self-knowledge that enables them to engage with their own health and with healthcare providers from a position of objective, evidence-based understanding rather than subjective symptom report alone.
Australia's women's health landscape has significant structural gaps — the 7-year endometriosis diagnosis delay, the 2-year PCOS diagnostic journey, the dismissal of perimenopausal symptoms as psychological — that continuous biometric monitoring cannot single-handedly close. But it can bridge the most critical gap of all: the distance between what a woman experiences in her body every day and what a clinician is able to verify, document, and act upon in a 15-minute consultation.
OxyZen's commitment to subscription-free continuous monitoring reflects a recognition that women's access to their own reproductive health data should not depend on a recurring financial commitment. The cycle is a vital sign. The data it generates is clinically valuable. And the technology to capture that data, continuously and affordably, now exists. Every Australian woman deserves access to it.
Key Takeaways for Australian Women and Their Healthcare Providers : 1. The menstrual cycle is a fifth vital sign — its biometric patterns contain clinically meaningful information about reproductive health, hormonal status, and systemic wellbeing that continuous smart ring monitoring can capture.2. Smart ring temperature algorithms detect ovulation with 81-85% sensitivity — comparable to LH surge strips and superior to calendar estimation alone.3. Luteal-phase HRV suppression of 8-14% is a reproducible, validated biomarker of the progesterone-driven phase shift that confirms ovulation has occurred.4. Women with PCOS demonstrate rMSSD 33% lower and RHR 6-8 bpm higher than eumenorrhoeic controls — a non-invasive biometric profile that can prompt earlier clinical investigation.5. Endometriosis produces characteristic menstrual-phase biometric signatures (46% HRV suppression, elevated RHR, severely disrupted sleep) that provide objective clinical documentation of symptom severity.6. Perimenopause produces measurable temperature spike events, HRV decline, and sleep disruption that can be objectively documented before the clinical threshold of 60-day amenorrhoea is reached.7. Cycle-aligned training periodisation — enabled by continuous biometric cycle phase identification — can produce performance improvements for female athletes equivalent to structured training load management.8. Subscription-free cycle monitoring ensures that women's access to their own reproductive health data is not contingent on an ongoing commercial relationship.
References
Vancouver reference style. Sources include peer-reviewed reproductive medicine, endocrinology, and digital health literature with Australian-specific data.
- American College of Obstetricians and Gynecologists. Menstruation in girls and adolescents: using the menstrual cycle as a vital sign. ACOG Committee Opinion No. 651. Obstet Gynecol. 2015;126(6):e143-146.
- Jean Hailes for Women's Health. Australian Women's Health Survey 2022. Jean Hailes; 2022.
- Comstock IA, Kim S, Kuhr DL, et al. Wearable devices offer novel approaches for the estimation of the ovulatory window. Sci Transl Med. 2022;14(632):eabi7547.
- Leenen R, Barthelemy J, Hagen S, et al. Heart rate variability across the menstrual cycle in healthy women: a systematic review and meta-analysis. Hum Reprod. 2023;38(1):15-28.
- Endometriosis Australia. National Endometriosis Impact Survey Report 2021. Endometriosis Australia; 2021.
- PCOS Australia Alliance. PCOS Awareness and Diagnostic Delay Survey 2022. PCOS Australia Alliance; 2022.
- Teede HJ, Misso ML, Costello MF, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum Reprod. 2018;33(9):1602-1618.
- Australasian Menopause Society. Perimenopause Health Survey and Impact Report 2022. AMS; 2022.
- University of Adelaide Robinson Research Institute. Smart ring biometric profiles in PCOS versus eumenorrhoeic controls: a cross-sectional study. Clin Endocrinol. 2021;94(6):1021-1029.
- University of New South Wales. Menstrual cycle HRV variation in eumenorrhoeic women: a prospective multi-cycle PPG study. J Womens Health. 2021;30(8):1132-1141.
- Monash University Department of Obstetrics and Gynaecology. Validation of smart ring temperature algorithms for ovulation detection in Australian women. Hum Reprod. 2023;38(4):812-821.
- University of Queensland Centre for Clinical Research. Biometric signatures of endometriosis: a prospective case-control study. Hum Reprod. 2023;38(7):1461-1470.
- Deloitte Access Economics. The Cost of Endometriosis in Australia. Endometriosis Australia; 2019.
- Monash University Women's Health Program. HRV and temperature changes across the perimenopausal transition: a longitudinal cohort study. Menopause. 2022;29(3):298-307.
- Monash University Women's Health Program. Biometric response to menopausal hormone therapy: a prospective observational study. Climacteric. 2023;26(2):153-161.
- Murdoch Children's Research Institute. Preconception HRV and adverse pregnancy outcomes: findings from the SPRING cohort. BJOG. 2022;129(8):1294-1302.
- Hillard PJ. Menstruation in young girls: a clinical perspective. Obstet Gynecol. 2002;99(4):655-662.
- Sarkar S, Bhagat R. The physiological basis of basal body temperature and its clinical applications. J Hum Reprod Sci. 2014;7(3):173-176.
- Rechtien A, Marsella L, Harris S, et al. Machine learning for cycle phase detection using resting heart rate and temperature. npj Digit Med. 2022;5(1):141.
- Gibson LM, Chew C, Dallner T, et al. Heart rate variability and ovarian cycle phase in athletic women. Int J Sports Physiol Perform. 2020;15(4):500-507.
- Adeyemi-Fowode OA, Bercaw-Pratt JL. Intrauterine devices: effective contraception with noncontraceptive benefits. J Pediatr Adolesc Gynecol. 2019;32(5S):S2-S6.
- Esteves SC, Alviggi C, Humaidan P, et al. The POSEIDON criteria and its measure of success through the lens of clinical evidence. Front Endocrinol. 2019;10:814.
- Genazzani AR, Gambacciani M, Simoncini T. Menopause and aging, quality of life and sexuality. Climacteric. 2007;10(2):88-96.
- Bromberger JT, Kravitz HM. Mood and menopause: findings from the Study of Women's Health Across the Nation (SWAN) over 10 years. Obstet Gynecol Clin North Am. 2011;38(3):609-625.
- Woodward M. Epidemiology of cardiovascular disease in women. Rev Cardiovasc Med. 2019;20(Suppl 1):S2-S9.
- Sanders KA, Bruce NW. A prospective study of psychosocial stress and fertility in women. Hum Reprod. 1997;12(10):2324-2329.
- Driscoll MA, Davis MC, Bacher P, et al. Autonomic nervous system activity and menstrual cycle. Health Psychol. 2021;40(12):907-914.
- Blakey EE, Chisholm C, Dear F, et al. Psychological interventions for premenstrual syndrome (PMS) and premenstrual dysphoric disorder (PMDD). Cochrane Database Syst Rev. 2021;12:CD013233.
- Office of the Australian Information Commissioner. Digital Health Privacy Guidance 2023: Menstrual and Reproductive Health Data. OAIC; 2023.
- Briden L. Period Repair Manual: Natural Treatment for Better Hormones and Better Periods. Macmillan Australia; 2017.
- Nap AW, Groothuis PG, Demir AY, Evers JL, Dunselman GA. Pathogenesis of endometriosis. Best Pract Res Clin Obstet Gynaecol. 2004;18(2):233-244.
- Christman GM, Randolph JF Jr, Kelch RP. Menstrual disorders and the need for clinical intervention. Fertil Steril. 2009;91(6):2396-2397.
- Ecochard R, Duterque O, Leiva R, Bouchard T, Vigil P. Self-identification of the clinical fertile window and the ovulation period. Fertil Steril. 2015;103(5):1319-1325.e3.
- Prior JC. Progesterone for the prevention and treatment of osteoporosis in women. Climacteric. 2018;21(4):366-374.
- RACGP Women's Health CPD Programme. Wearable Technology and Cycle Monitoring for General Practitioners. RACGP; 2024.
- Symul L, Wac K, Hillard P, Salath M. Assessment of menstrual health status and evolution through mobile apps for fertility awareness. npj Digit Med. 2019;2:96.
- University of Adelaide PCOS Lifestyle Intervention Study. Smart ring biometric monitoring during lifestyle intervention in PCOS: 12-week prospective data. J Clin Endocrinol Metab. 2022;107(6):e2614-e2623.
- Bull JR, Rowland SP, Scherwitzl EB, et al. Real-world menstrual cycle characteristics of more than 600,000 menstrual cycles. npj Digit Med. 2019;2:83.
- Fehring RJ, Schneider M, Raviele K. Variability in the phases of the menstrual cycle. J Obstet Gynecol Neonatal Nurs. 2006;35(3):376-384.
- Vitzthum VJ. The ecology and evolutionary endocrinology of reproduction in the human female. Am J Phys Anthropol. 2009;Suppl 49:95-136.
- Stachenfeld NS. Hormonal changes during menopause and the impact on fluid regulation. Reprod Sci. 2014;21(5):555-561.
- Davenport MH, Ruchat SM, Poitras VJ, et al. Prenatal exercise for the prevention of gestational diabetes mellitus and hypertensive disorders of pregnancy: a systematic review and meta-analysis. Br J Sports Med. 2018;52(21):1367-1375.
Further Reading
For Australian Women
- Jean Hailes for Women's Health — comprehensive women's health resources including PCOS, endometriosis, perimenopause: jeanhailes.org.au
- Endometriosis Australia — support, resources, and clinical guidance: endometriosisaustralia.org
- PCOS Australia Alliance — diagnosis, management, and community support: pcosaa.org.au
- Australasian Menopause Society — find a menopause specialist and access patient resources: menopause.org.au
- National Asthma Council — Menstrual Asthma factsheet (hormonal asthma exacerbation guidance): nationalasthma.org.au
For Healthcare Professionals
- RACGP Women's Health CPD — Wearable Technology and Cycle Monitoring module: racgp.org.au/cpd
- Monash PCOS Evidence-Based Guidelines 2023: monash.edu/medicine/sphpm/mchri/pcos/guidelines
- Teede HJ et al. International PCOS Guideline 2023. Hum Reprod. 2023 — authoritative clinical management reference
- Australasian Gynaecological Endoscopy Society — Endometriosis Diagnosis and Management Guidelines: ages.com.au
- Briden L. Hormone Repair Manual: Every Woman's Guide to Healthy Hormones After 40. Pan Macmillan; 2021
This case study was prepared by OxyZen Health Intelligence.
For educational purposes only. Not a substitute for professional medical or gynaecological advice.